Alginate-Bentonite Encapsulation of Extremophillic Bacterial Consortia Enhances Chenopodium quinoa Tolerance to Metal Stress
- PMID: 39458375
- PMCID: PMC11509983
- DOI: 10.3390/microorganisms12102066
Alginate-Bentonite Encapsulation of Extremophillic Bacterial Consortia Enhances Chenopodium quinoa Tolerance to Metal Stress
Erratum in
-
Correction: Alvarado et al. Alginate-Bentonite Encapsulation of Extremophillic Bacterial Consortia Enhances Chenopodium quinoa Tolerance to Metal Stress. Microorganisms 2024, 12, 2066.Microorganisms. 2024 Nov 19;12(11):2356. doi: 10.3390/microorganisms12112356. Microorganisms. 2024. PMID: 39597771 Free PMC article.
Abstract
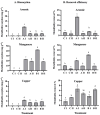
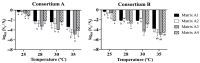
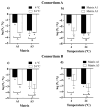
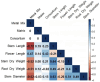
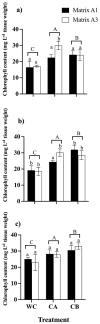
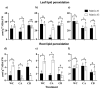
This study explores the encapsulation in alginate/bentonite beads of two metal(loid)-resistant bacterial consortia (consortium A: Pseudomonas sp. and Bacillus sp.; consortium B: Pseudomonas sp. and Bacillus sp.) from the Atacama Desert (northern Chile) and Antarctica, and their influence on physiological traits of Chenopodium quinoa growing in metal(loid)-contaminated soils. The metal(loid) sorption capacity of the consortia was determined. Bacteria were encapsulated using ionic gelation and were inoculated in soil of C. quinoa. The morphological variables, photosynthetic pigments, and lipid peroxidation in plants were evaluated. Consortium A showed a significantly higher biosorption capacity than consortium B, especially for As and Cu. The highest viability of consortia was achieved with matrices A1 (3% alginate and 2% bentonite) and A3 (3% alginate, 2% bentonite and 2.5% LB medium) at a drying temperature of 25 °C and storage at 4 °C. After 12 months, the highest viability was detected using matrix A1 with a concentration of 106 CFU g-1. Further, a greenhouse experiment using these consortia in C. quinoa plants showed that, 90 days after inoculation, the morphological traits of both consortia improved. Chemical analysis of metal(loid) contents in the leaves indicated that consortium B reduced the absorption of Cu to 32.1 mg kg-1 and that of Mn to 171.9 mg kg-1. Encapsulation resulted in a significant increase in bacterial survival. This highlights the benefits of using encapsulated microbial consortia from extreme environments, stimulating the growth of C. quinoa, especially in soils with metal(loid) levels that can be a serious constraint for plant growth.
Keywords: bacterial storage; extremophilic bacteria; metal(loid)s biosorption; metal(loid)s toxicity alleviation.
Conflict of interest statement
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures
References
-
- Angeli V., Silva P., Massuela D., Khan M.W., Hamar A., Khajehei F., Graeff-Hönninger S., Piatti C. Quinoa (Chenopodium quinoa Willd.): An Overview of the Potentials of the “Golden Grain” and Socio-Economic and Environmental Aspects of Its Cultivation and Marketization. Foods. 2020;9:216. doi: 10.3390/foods9020216. - DOI - PMC - PubMed
-
- Yang A., Akhtar S., Amjad M., Iqbal S., Jacobsen S.E. Growth and physiological responses of quinoa to drought and temperature stress. J. Agron. Crop Sci. 2016;202:445–453. doi: 10.1111/jac.12167. - DOI
-
- Bhargava A., Shukla S., Srivastava J., Singh N., Ohri D. Chenopodium: A prospective plant for phytoextraction. Acta Physiol. Plants. 2008;30:111–120. doi: 10.1007/s11738-007-0097-3. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

