PD-L1 blockade immunotherapy rewires cancer-induced emergency myelopoiesis
- PMID: 39464894
- PMCID: PMC11502414
- DOI: 10.3389/fimmu.2024.1386838
PD-L1 blockade immunotherapy rewires cancer-induced emergency myelopoiesis
Abstract
Introduction: Immune checkpoint blockade (ICB) immunotherapy has revolutionized cancer treatment, demonstrating exceptional clinical responses in a wide range of cancers. Despite the success, a significant proportion of patients still fail to respond, highlighting the existence of unappreciated mechanisms of immunotherapy resistance. Delineating such mechanisms is paramount to minimize immunotherapy failures and optimize the clinical benefit.
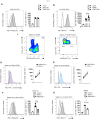
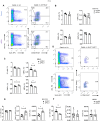
Methods: In this study, we treated tumour-bearing mice with PD-L1 blockage antibody (aPD-L1) immunotherapy, to investigate its effects on cancer-induced emergency myelopoiesis, focusing on bone marrow (BM) hematopoietic stem and progenitor cells (HSPCs). We examined the impact of aPD-L1 treatment on HSPC quiescence, proliferation, transcriptomic profile, and functionality.
Results: Herein, we reveal that aPD-L1 in tumour-bearing mice targets the HSPCs in the BM, mediating their exit from quiescence and promoting their proliferation. Notably, disruption of the PDL1/PD1 axis induces transcriptomic reprogramming in HSPCs, observed in both individuals with Hodgkin lymphoma (HL) and tumour-bearing mice, shifting towards an inflammatory state. Furthermore, HSPCs from aPDL1-treated mice demonstrated resistance to cancer-induced emergency myelopoiesis, evidenced by a lower generation of MDSCs compared to control-treated mice.
Discussion: Our findings shed light on unrecognized mechanisms of action of ICB immunotherapy in cancer, which involves targeting of BM-driven HSPCs and reprogramming of cancer-induced emergency myelopoiesis.
Keywords: bone marrow; cancer; hematopoietic stem and progenitor cell; immunotherapy; inflammation.
Copyright © 2024 Boumpas, Papaioannou, Bousounis, Grigoriou, Bergo, Papafragkos, Tasis, Iskas, Boon, Makridakis, Vlachou, Gavriilaki, Hatzioannou, Mitroulis, Trompouki and Verginis.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures





References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

