Catalytic Asymmetric Diastereodivergent Synthesis of 2-Alkenylindoles Bearing both Axial and Central Chirality
- PMID: 39474410
- PMCID: PMC11504580
- DOI: 10.1021/prechem.4c00008
Catalytic Asymmetric Diastereodivergent Synthesis of 2-Alkenylindoles Bearing both Axial and Central Chirality
Abstract
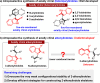
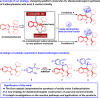
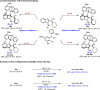
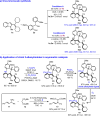
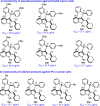
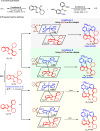
The catalytic asymmetric diastereodivergent synthesis of axially chiral 2-alkenylindoles was established via chiral phosphoric acid-catalyzed addition reactions of C3-unsubstituted 2-alkenylindoles with o-hydroxybenzyl alcohols under different reaction conditions. Using this strategy, two series of 2-alkenylindoles bearing both axial and central chirality were synthesized in a diastereodivergent fashion with moderate to high yields and good stereoselectivities (up to 99% yield, 95:5 er, >95:5 dr). Moreover, theoretical calculations were performed on the key transition states leading to different stereoisomers, which provided an in-depth understanding of the origin of the observed stereoselectivity and diastereodivergence of the products under different reaction conditions. More importantly, these 2-alkenylindoles were utilized in asymmetric catalysis as chiral organocatalysts and in medicinal chemistry for evaluation of their cytotoxicity, which demonstrated their potential applications. This study has not only established the catalytic atroposelective synthesis of axially chiral 2-alkenylindoles, but also provided an efficient strategy for catalytic asymmetric diastereodivergent construction of indole-based scaffolds bearing both axial and central chirality.
© 2024 The Authors. Co-published by University of Science and Technology of China and American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures



Similar articles
-
Design and Organocatalytic Asymmetric Synthesis of Indolyl-Pyrroloindoles Bearing Both Axial and Central Chirality.J Org Chem. 2023 Jun 16;88(12):7684-7702. doi: 10.1021/acs.joc.2c02303. Epub 2022 Nov 23. J Org Chem. 2023. PMID: 36417567
-
Organocatalytic Atroposelective Synthesis of Indole Derivatives Bearing Axial Chirality: Strategies and Applications.Acc Chem Res. 2022 Sep 20;55(18):2562-2580. doi: 10.1021/acs.accounts.2c00465. Epub 2022 Sep 2. Acc Chem Res. 2022. PMID: 36053083
-
Design and synthesis of axially chiral aryl-pyrroloindoles via the strategy of organocatalytic asymmetric (2 + 3) cyclization.Fundam Res. 2022 Jan 21;3(2):237-248. doi: 10.1016/j.fmre.2022.01.002. eCollection 2023 Mar. Fundam Res. 2022. PMID: 38932922 Free PMC article.
-
A Strategy for Synthesizing Axially Chiral Naphthyl-Indoles: Catalytic Asymmetric Addition Reactions of Racemic Substrates.Angew Chem Int Ed Engl. 2019 Oct 14;58(42):15104-15110. doi: 10.1002/anie.201908279. Epub 2019 Sep 10. Angew Chem Int Ed Engl. 2019. PMID: 31441203 Review.
-
Recent advances in the asymmetric phosphoric acid-catalyzed synthesis of axially chiral compounds.Beilstein J Org Chem. 2021 Nov 15;17:2729-2764. doi: 10.3762/bjoc.17.185. eCollection 2021. Beilstein J Org Chem. 2021. PMID: 34876929 Free PMC article. Review.
Cited by
-
Atroposelective construction of axially chiral alkenylindole-fused nine-membered rings via catalytic asymmetric formal (4 + 5) cycloaddition.Nat Commun. 2025 Jul 17;16(1):6605. doi: 10.1038/s41467-025-62035-y. Nat Commun. 2025. PMID: 40676028 Free PMC article.
-
Chiral phosphoric acid-catalyzed atroposelective iodination of N-arylindoles.Commun Chem. 2025 Jun 21;8(1):190. doi: 10.1038/s42004-025-01584-1. Commun Chem. 2025. PMID: 40544218 Free PMC article.
-
Stereoselective synthesis of atropisomeric amides enabled by intramolecular acyl transfer.Chem Sci. 2025 Jan 23;16(9):3938-3945. doi: 10.1039/d4sc05760k. eCollection 2025 Feb 26. Chem Sci. 2025. PMID: 39886439 Free PMC article.
References
-
- Foye’s Principles of Medicinal Chemistry, 7th ed.; Lemke T. L., Williams D. A., Roche V. F., Zito S. W., Eds.; Lippincott Williams & Wilkins, Wolters Kluwer: Baltimore, 2013.
LinkOut - more resources
Full Text Sources
Miscellaneous
