Decisive role of mDia-family formins in cell cortex function of highly adherent cells
- PMID: 39475610
- PMCID: PMC11524191
- DOI: 10.1126/sciadv.adp5929
Decisive role of mDia-family formins in cell cortex function of highly adherent cells
Abstract
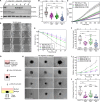
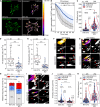
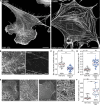
Cortical formins, pivotal for the assembly of linear actin filaments beneath the membrane, exert only minor effects on unconfined cell migration of weakly and moderately adherent cells. However, their impact on migration and mechanostability of highly adherent cells remains poorly understood. Here, we demonstrate that loss of cortical actin filaments generated by the formins mDia1 and mDia3 drastically compromises cell migration and mechanics in highly adherent fibroblasts. Biophysical analysis of the mechanical properties of the mutant cells revealed a markedly softened cell cortex in the poorly adherent state. Unexpectedly, in the highly adherent state, associated with a hyperstretched morphology with exaggerated focal adhesions and prominent high-strain stress fibers, they exhibited even higher cortical tension compared to control. Notably, misguidance of intracellular forces, frequently accompanied by stress-fiber rupture, culminated in the formation of tension- and contractility-induced macroapertures, which was instantly followed by excessive lamellipodial protrusion at the periphery, providing critical insights into mechanotransduction of mechanically stressed and highly adherent cells.
Figures





References
-
- Blanchoin L., Boujemaa-Paterski R., Sykes C., Plastino J., Actin dynamics, architecture, and mechanics in cell motility. Physiol. Rev. 94, 235–263 (2014). - PubMed
-
- Salbreux G., Charras G. T., Paluch E. K., Actin cortex mechanics and cellular morphogenesis. Trends Cell Biol. 22, 536–545 (2012). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

