Reliable replicative lifespan determination of yeast with a single-channel microfluidic chip
- PMID: 39479938
- PMCID: PMC11657196
- DOI: 10.1242/bio.060596
Reliable replicative lifespan determination of yeast with a single-channel microfluidic chip
Abstract
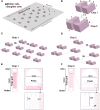
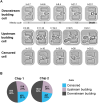
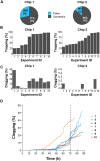
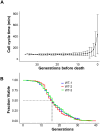
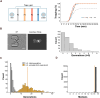
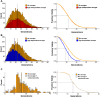
Saccharomyces cerevisiae is a powerful model for aging research due to its short lifespan and genetic malleability. Microfluidic devices offer an attractive approach enabling rapid monitoring of hundreds of cells during their entire replicative lifespan (RLS). Yet, key operational issues such as contaminations, cell loss, and cell-aggregates-dependent flow obstruction can hinder RLS experiments. We report the development of a microfluidic device configuration that effectively prevents flow blockage. We conducted comprehensive performance characterization, evaluating trapping efficiency, cell retention, budding orientation, and cell aggregate formation. The optimized device successfully supported long-term culturing and reliable RLS measurements of budding yeast strains. For accurate lifespan determination, a detailed workflow is provided that includes device fabrication, live microscopy setup, and characterization of cell age distribution. This work describes an accessible and reliable microfluidic device for yeast RLS studies, promoting further exploration in aging research.
Keywords: Clogging mitigation; Microfluidics; Replicative lifespan.
© 2024. Published by The Company of Biologists Ltd.
Conflict of interest statement
Competing interests The authors declare no competing or financial interests.
Figures

Similar articles
-
A high-throughput microfluidic diploid yeast long-term culturing (DYLC) chip capable of bud reorientation and concerted daughter dissection for replicative lifespan determination.J Nanobiotechnology. 2022 Mar 31;20(1):171. doi: 10.1186/s12951-022-01379-9. J Nanobiotechnology. 2022. PMID: 35361237 Free PMC article.
-
Measuring the Replicative Lifespan of Saccharomyces cerevisiae Using the HYAA Microfluidic Platform.Methods Mol Biol. 2020;2144:1-6. doi: 10.1007/978-1-0716-0592-9_1. Methods Mol Biol. 2020. PMID: 32410020 Free PMC article.
-
Investigation of daughter cell dissection coincidence of single budding yeast cells immobilized in microfluidic traps.Anal Bioanal Chem. 2021 Mar;413(8):2181-2193. doi: 10.1007/s00216-021-03186-x. Epub 2021 Jan 31. Anal Bioanal Chem. 2021. PMID: 33517467
-
Microfluidic technologies for yeast replicative lifespan studies.Mech Ageing Dev. 2017 Jan;161(Pt B):262-269. doi: 10.1016/j.mad.2016.03.009. Epub 2016 Mar 23. Mech Ageing Dev. 2017. PMID: 27015709 Free PMC article. Review.
-
Microfluidic Based Optical Microscopes on Chip.Cytometry A. 2018 Oct;93(10):987-996. doi: 10.1002/cyto.a.23589. Epub 2018 Sep 13. Cytometry A. 2018. PMID: 30211977 Free PMC article. Review.
References
-
- Crane, M. M., Russell, A. E., Schafer, B. J., Blue, B. W., Whalen, R., Almazan, J., Hong, M. G., Nguyen, B., Goings, J. E., Chen, K. L.et al. (2019). DNA damage checkpoint activation impairs chromatin homeostasis and promotes mitotic catastrophe during aging. Elife 8, e50778. 10.7554/eLife.50778 - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

