Endothelial cell-specific progerin expression does not cause cardiovascular alterations and premature death
- PMID: 39479939
- PMCID: PMC11822624
- DOI: 10.1111/acel.14389
Endothelial cell-specific progerin expression does not cause cardiovascular alterations and premature death
Abstract
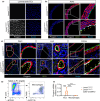
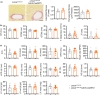
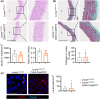
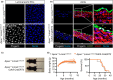
Hutchinson-Gilford progeria syndrome (HGPS) is a rare genetic disorder caused by a mutation in the LMNA gene that provokes the synthesis of progerin, a mutant version of the nuclear protein lamin A that accelerates aging and precipitates death. The most clinically relevant feature of HGPS is the development of cardiac anomalies and severe vascular alterations, including massive loss of vascular smooth muscle cells, increased fibrosis, and generalized atherosclerosis. However, it is unclear if progerin expression in endothelial cells (ECs) causes the cardiovascular manifestations of HGPS. To tackle this question, we generated atherosclerosis-free mice (LmnaLCS/LCSCdh5-CreERT2) and atheroprone mice (Apoe-/-LmnaLCS/LCSCdh5-CreERT2) with EC-specific progerin expression. Like progerin-free controls, LmnaLCS/LCSCdh5-CreERT2 mice did not develop heart fibrosis or cardiac electrical and functional alterations, and had normal vascular structure, body weight, and lifespan. Similarly, atheroprone Apoe-/-LmnaLCS/LCSCdh5-CreERT2 mice showed no alteration in body weight or lifespan versus Apoe-/-LmnaLCS/LCS controls and did not develop vascular alterations or aggravated atherosclerosis. Our results indicate that progerin expression in ECs is not sufficient to cause the cardiovascular phenotype and premature death associated with progeria.
Keywords: Hutchinson‐Gilford progeria syndrome; atherosclerosis; cardiovascular disease; endothelial cells; progerin.
© 2024 The Author(s). Aging Cell published by Anatomical Society and John Wiley & Sons Ltd.
Conflict of interest statement
All the contributing authors declared no conflicts of interest. The funders had no role in the design of the study, the collection, analysis, or interpretation of data, and reporting of the study.
Figures


Similar articles
-
Vascular Smooth Muscle-Specific Progerin Expression Accelerates Atherosclerosis and Death in a Mouse Model of Hutchinson-Gilford Progeria Syndrome.Circulation. 2018 Jul 17;138(3):266-282. doi: 10.1161/CIRCULATIONAHA.117.030856. Epub 2018 Feb 28. Circulation. 2018. PMID: 29490993 Free PMC article.
-
Exacerbated atherosclerosis in progeria is prevented by progerin elimination in vascular smooth muscle cells but not endothelial cells.Proc Natl Acad Sci U S A. 2024 Apr 30;121(18):e2400752121. doi: 10.1073/pnas.2400752121. Epub 2024 Apr 22. Proc Natl Acad Sci U S A. 2024. PMID: 38648484 Free PMC article.
-
Vascular smooth muscle cell-specific progerin expression in a mouse model of Hutchinson-Gilford progeria syndrome promotes arterial stiffness: Therapeutic effect of dietary nitrite.Aging Cell. 2019 Jun;18(3):e12936. doi: 10.1111/acel.12936. Epub 2019 Mar 18. Aging Cell. 2019. PMID: 30884114 Free PMC article.
-
Vascular smooth muscle cell loss underpins the accelerated atherosclerosis in Hutchinson-Gilford progeria syndrome.Nucleus. 2019 Dec;10(1):28-34. doi: 10.1080/19491034.2019.1589359. Nucleus. 2019. PMID: 30900948 Free PMC article. Review.
-
Molecular and Cellular Mechanisms Driving Cardiovascular Disease in Hutchinson-Gilford Progeria Syndrome: Lessons Learned from Animal Models.Cells. 2021 May 11;10(5):1157. doi: 10.3390/cells10051157. Cells. 2021. PMID: 34064612 Free PMC article. Review.
References
-
- Abutaleb, N. O. , Atchison, L. , Choi, L. , Bedapudi, A. , Shores, K. , Gete, Y. , Cao, K. , & Truskey, G. A. (2023). Lonafarnib and everolimus reduce pathology in iPSC‐derived tissue engineered blood vessel model of Hutchinson‐Gilford progeria syndrome. Scientific Reports, 13(1), 5032. 10.1038/s41598-023-32035-3 - DOI - PMC - PubMed
-
- Atchison, L. , Abutaleb, N. O. , Snyder‐Mounts, E. , Gete, Y. , Ladha, A. , Ribar, T. , Cao, K. , & Truskey, G. A. (2020). iPSC‐derived endothelial cells affect vascular function in a tissue‐engineered blood vessel model of Hutchinson‐Gilford progeria syndrome. Stem Cell Reports, 14(2), 325–337. 10.1016/j.stemcr.2020.01.005 - DOI - PMC - PubMed
-
- Benedicto, I. , Carmona, R. M. , Barettino, A. , Espinós‐Estévez, C. , Gonzalo, P. , Nevado, R. M. , de la Fuente‐Pérez, M. , Andrés‐Manzano, M. J. , González‐Gómez, C. , Rolas, L. , Dorado, B. , Nourshargh, S. , Hamczyk, M. R. , & Andrés, V. (2024). Exacerbated atherosclerosis in progeria is prevented by progerin elimination in vascular smooth muscle cells but not endothelial cells. Proceedings of the National Academy of Sciences, 121(18), e2400752121. 10.1073/pnas.2400752121 - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
- BES-2017-079705/Ministerio de Ciencia, Innovación y Universidades
- CEX2020-001041-S/Ministerio de Ciencia, Innovación y Universidades
- IJC2019-040798-I/Ministerio de Ciencia, Innovación y Universidades
- PID2022-137111OA-I00/Ministerio de Ciencia, Innovación y Universidades
- PID2022-141211OB-I00/Ministerio de Ciencia, Innovación y Universidades
- RYC2021-033805-I/Ministerio de Ciencia, Innovación y Universidades
- European Union NextGenerationEU/PRTR
- Instituto de Salud Carlos III
- European Regional Development Fund
- European Structural and Investment Funds
- Pro-CNIC Foundation
- 2017-T1/BMD-5247/Comunidad de Madrid
- 2021-5A/BMD-20944/Comunidad de Madrid
- FPU16/05027/Ministerio de Educación, Cultura y Deporte
- LCF/BQ/DR19/1170012/'la Caixa' Foundation
LinkOut - more resources
Full Text Sources
Miscellaneous

