Unveiling the antimetastatic activity of monoterpene indole alkaloids targeting MMP9 in cancer cells, with a focus on pharmacokinetic and cellular insights
- PMID: 39481856
- PMCID: PMC11625137
- DOI: 10.1016/j.mocell.2024.100143
Unveiling the antimetastatic activity of monoterpene indole alkaloids targeting MMP9 in cancer cells, with a focus on pharmacokinetic and cellular insights
Abstract
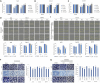
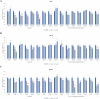
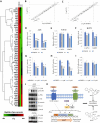
Distant metastasis, together with acquired resistance, limits the therapeutic impact of chemotherapy and molecularly targeted therapies. The properties of the tumor microenvironment determine how sensitive or resistant various cancers are to specific pharmacological treatments. Matrix metalloproteinase 9 (MMP9) is widely known for its ability to break down the extracellular matrix and it also modulates the motility of cancer cells. Here, our goal was to identify compounds that target MMP9 and evaluate their capacity to inhibit the motility of cancer cells. The antimetastatic effect of monoterpene indole alkaloids (MIAs) on cell viability and motility was evaluated by methyl thiazolyl tetrazolium assay, migration assay, invasion assay, quantitative real-time polymerase chain reaction, pathway-focused expression analysis, Western blotting, reporter assay, molecular docking simulation, and target prediction. MIA compounds target MMP9. MIAs inhibited the expression of phospho-epidermal growth factor receptor, phospho-Akt, phospho-JNK, and cyclin D1. Additionally, MIAs had predicted favorable pharmacokinetic profile and drug-like properties. Furthermore, among the MIA compounds, lyaloside and 5(S)-5-carbomethoxystrictosidine had low cytotoxicity and regulated cancer-related signaling, including cell migration, cell invasion, epithelial-mesenchymal transition, and immune evasion. Our findings demonstrated that the MIAs used in this study have potential antimetastasis properties that occur via MMP9-mediated regulation of cancer signaling and have the potential to be used therapeutically at safe doses.
Keywords: 5(S)-5-carbomethoxystrictosidine; Anticancer; Lyaloside; Matrix metalloproteinase 9; Monoterpene indole alkaloids.
Copyright © 2024 The Authors. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of Competing Interests The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures


References
-
- Afza N., Fatma S., Ghous F., Shukla S., Rai S., Srivastava K., Bishnoi A. An efficient multicomponent synthesis, characterization, SAR, In-silico ADME prediction and molecular docking studies of 2-Amino-7-(substituted-phenyl)-3-cyano-4-phenyl-4,5,6,7-tetrahydropyrano[2,3-b] pyrrole-5-carboxylic acid derivatives and their in-vitro antimicrobial activity. J. Mol. Struct. 2023;1276 doi: 10.1016/J.MOLSTRUC.2022.134721. - DOI
-
- Bouzina A., Bouone Y.O., Sekiou O., Aissaoui M., Ouk T.-S., Djemel A., Mansouri R., Ibrahim-Ouali M., Bouslama Z., Aouf N.E. In vitro antitumor activity, molecular dynamics simulation, DFT study, ADME prediction, and Eg5 binding of enastron analogues. RSC Adv. 2023;13(28):19567–19584. doi: 10.1039/D3RA02904B. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

