Newborn screening for SCID: the very first prospective pilot study from Türkiye
- PMID: 39483481
- PMCID: PMC11526446
- DOI: 10.3389/fimmu.2024.1384195
Newborn screening for SCID: the very first prospective pilot study from Türkiye
Abstract
Purpose: The measurement of T-cell receptor excision circle (TREC) is used for newborn screening (NBS) in dried blood spot (DBS) samples from Guthrie card for severe combined immunodeficiency (SCID). Here, we report the results of first newborn screening pilot program for SCID conducted in Türkiye.
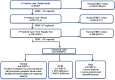
Methods: The study was carried out together with Ankara University School of Medicine and The Ministry of Health, Public Health General Directorate, Pediatric and Adolescent Health Department. TREC measurements were performed in randomly selected Guthrie card samples obtained from 20253 babies born between October 2018 and October 2020. The TREC analyses were performed together with beta Actin (β-Actin) via RT-PCR (Real Time Polymerase Chain Reaction).
Results: TRECs found to be normal (≥15 copies/µl) in 98,6% of the newborns (n: 19975) but low (<15 copies/µl) in 1.4% (n:278) at the initial analyses. TRECs were retested in 278 suspected infants and found to be normal in 160 (0.8%) while low in 118 (0.58%). New DBS were obtained from the babies with low TRECs (new sample test). TRECs were normal in 108 (0.53%) of the new sample tests and low in 10 (0.049%). Two among 10 babies who had abnormal (undetectable) TRECs were diagnosed as SCID; ADA (P1) and RAG1 (P2) defects were confirmed respectively. They both received curative treatments [gene therapy (P1) and HSCT (P2)]. The remaining 6 of 8 newborns with abnormal TRECs were found normal after clinical and laboratory immune work-up, while medical records of other two revealed early postnatal death due to extreme prematurity.
Conclusion: In the light of this study the incidence of SCID was detected at least 1/10000 live births in Türkiye. This study shows the feasibility and usefulness of initiating SCID screening in Türkiye.
Keywords: SCID; TREC; Türkiye; dried blood sample (DBS); newborn screening.
Copyright © 2024 Haskologlu, Kocak, Tufan, Aksoy, Bastug, Oner, Islamoglu, Baskin, Esenboga, Acican, Ceylaner, Guner, Keles, Cagdas, Reisli, Tezel, Dogu, Tezcan and Ikinciogullari.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous