The Effects of Zinc on Proprioceptive Sensory Function and Nerve Conduction
- PMID: 39484175
- PMCID: PMC11523716
- DOI: 10.3390/neurosci4040025
The Effects of Zinc on Proprioceptive Sensory Function and Nerve Conduction
Abstract
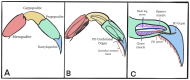
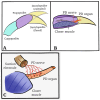
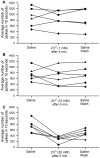
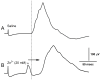
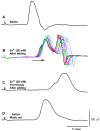
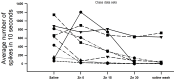
Zinc (Zn2+) is an essential element that can promote proper organ function, cell growth, and immune response; it can also, however, be present in too great a quantity. Zinc toxicity caused by overexposure may result in both minor and major physiological effects, with chronic exposure at low levels and acute exposure at high levels being harmful or even toxic. This investigation examines the effects of acute exposure to relatively high concentrations of Zn2+ on sensory nerve function and nerve conduction. A proprioceptive nerve in marine crab (Callinectes sapidus) limbs was used as a model to assess the effects of Zn2+ on stretch-activated channels (SACs) and evoked nerve conduction. Exposure to Zn2+ slowed nerve condition rapidly; however, several minutes were required before the SACs in sensory endings were affected. A depression in conduction speed and an increase followed by a decrease in amplitude were observed for the evoked compound action potential, while the frequency of nerve activity upon joint movement and stretching of the chordotonal organ significantly decreased. These altered responses could be partially reversed via extensive flushing with fresh saline to remove the zinc. This indicates that subtle, long-term exposure to Zn2+ may alter an organism's SAC function for channels related to proprioception and nerve conduction.
Keywords: conduction; crustacean; proprioception; recruitment; sensory; zinc.
© 2023 by the authors.
Conflict of interest statement
Conflicts of InterestThe authors declare no conflict of interest.
Figures


References
-
- Ryu M.-S., Aydemir T.B. Zinc. In: Marriott B.P., Birt D.F., Stallings V.A., Yates A.A., editors. Present Knowledge in Nutrition. 11th ed. Wiley-Blackwell; Cambridge, MA, USA: 2020. pp. 393–408.
Grants and funding
LinkOut - more resources
Full Text Sources

