Transcriptomic alterations in APP/PS1 mice astrocytes lead to early postnatal axon initial segment structural changes
- PMID: 39485512
- PMCID: PMC11530419
- DOI: 10.1007/s00018-024-05485-9
Transcriptomic alterations in APP/PS1 mice astrocytes lead to early postnatal axon initial segment structural changes
Abstract
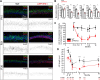
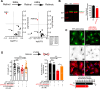
Alzheimer´s disease (AD) is characterized by neuronal function loss and degeneration. The integrity of the axon initial segment (AIS) is essential to maintain neuronal function and output. AIS alterations are detected in human post-mortem AD brains and mice models, as well as, neurodevelopmental and mental disorders. However, the mechanisms leading to AIS deregulation in AD and the extrinsic glial origin are elusive. We studied early postnatal differences in AIS cellular/molecular mechanisms in wild-type or APP/PS1 mice and combined neuron-astrocyte co-cultures. We observed AIS integrity alterations, reduced ankyrinG expression and shortening, in APP/PS1 mice from P21 and loss of AIS integrity at 21 DIV in wild-type and APP/PS1 neurons in the presence of APP/PS1 astrocytes. AnkyrinG decrease is due to mRNAs and protein reduction of retinoic acid synthesis enzymes Rdh1 and Aldh1b1, as well as ADNP (Activity-dependent neuroprotective protein) in APP/PS1 astrocytes. This effect was mimicked by wild-type astrocytes expressing ADNP shRNA. In the presence of APP/PS1 astrocytes, wild-type neurons AIS is recovered by inhibition of retinoic acid degradation, and Adnp-derived NAP peptide (NAPVSIPQ) addition or P2X7 receptor inhibition, both regulated by retinoic acid levels. Moreover, P2X7 inhibitor treatment for 2 months impaired AIS disruption in APP/PS1 mice. Our findings extend current knowledge on AIS regulation, providing data to support the role of astrocytes in early postnatal AIS modulation. In conclusion, AD onset may be related to very early glial cell alterations that induce AIS and neuronal function changes, opening new therapeutic approaches to detect and avoid neuronal function loss.
Keywords: ADNP; AnkyrinG; Astrocytes; Axon initial segment; Neurodegeneration; P2X7; Retinoic acid.
© 2024. The Author(s).
Conflict of interest statement
The authors have no relevant financial or non-financial interests to disclose.
Figures






References
-
- Crystal H, Dickson D, Fuld P, Masur D, Scott R, Mehler M, Masdeu J, Kawas C, Aronson M, Wolfson L (1988) Clinico-pathologic studies in dementia: nondemented subjects with pathologically confirmed Alzheimer’s disease. Neurology 38:1682–1687 - PubMed
-
- Harris SS, Wolf F, De Strooper B, Busche MA (2020) Tipping the scales: peptide-dependent dysregulation of neural circuit dynamics in Alzheimer’s disease. Neuron 107:417–435 - PubMed
-
- Maestu F, de Haan W, Busche MA, DeFelipe J (2021) Neuronal excitation/inhibition imbalance: core element of a translational perspective on Alzheimer pathophysiology. Ageing Res Rev 69:101372 - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

