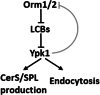
Orm proteins control ceramide synthesis and endocytosis via LCB-mediated Ypk1 regulation
- PMID: 39490931
- PMCID: PMC11621495
- DOI: 10.1016/j.jlr.2024.100683
Orm proteins control ceramide synthesis and endocytosis via LCB-mediated Ypk1 regulation
Abstract
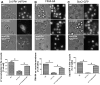
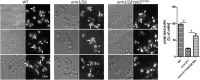
Sphingolipids (SPLs) are major components of cell membranes with significant functions. Their production is a highly-regulated multi-step process with the formation of two major intermediates, long chain bases (LCBs) and ceramides. Homologous Orm proteins in both yeast and mammals negatively regulate LCB production by inhibiting serine palmitoyltransferase (SPT), the first enzyme in SPL de novo synthesis. Orm proteins are therefore regarded as major regulators of SPL production. Combining targeted lipidomic profiling with phenotypic analysis of yeast mutants with both ORM1 and ORM2 deleted (orm1/2Δ), we report here that Ypk1, an AGC family protein kinase, signaling is compromised in an LCB-dependent manner. In orm1/2Δ, phosphorylation of Ypk1 at its activation sites is reduced, and so is its in vivo activity shown by reduced phosphorylation of Ypk1 substrate, Lac1, the catalytic component of ceramide synthase (CerS). A corresponding defect in ceramide synthesis was detected, preventing the extra LCBs generated in orm1/2Δ from fully converting into downstream SPL products. The results suggest that Orm proteins play a complex role in regulating SPL production in yeast S. cerevisiae by exerting an extra and opposite effect on CerS. Functionally, we define endocytosis and an actin polarization defect of orm1/2Δ and demonstrate the roles of Ypk1 in mediating the effects of Orm proteins on endocytosis. Collectively, the results reveal a previously unrecognized role of yeast Orm proteins in controlling ceramide synthesis and their function in endocytosis through regulating Ypk1 signaling.
Keywords: LCBs; Orm1/2; SPT; Ypk1; cell signaling; endocytosis; enzymology/enzyme regulation; lipidomics; lipids; sphingolipids.
Published by Elsevier Inc.
Conflict of interest statement
Conflict of interest The authors declare the following financial interests/personal relationships which may be considered as potential competing interests. Dr Maurizio Del Poeta, MD, is a Co-Founder and Chief Scientific Officer (CSO) of MicroRid Technologies Inc. The goal of MicroRid Technologies Inc is to develop new anti-fungal agents of therapeutic use. All other authors declare no competing interests.
Figures




References
-
- Dickson R.C., Lester R.L. Yeast sphingolipids. Biochim. Biophys. Acta. 1999;1426:347–357. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

