Automated and closed clinical-grade manufacturing protocol produces potent NK cells against neuroblastoma cells and AML blasts
- PMID: 39496674
- PMCID: PMC11535237
- DOI: 10.1038/s41598-024-76791-2
Automated and closed clinical-grade manufacturing protocol produces potent NK cells against neuroblastoma cells and AML blasts
Abstract
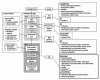
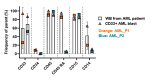
Natural killer (NK) cells are a promising allogeneic immunotherapy option due to their natural ability to kill tumor cells, and due to their apparent safety. This study describes the development of a GMP-compliant manufacturing protocol for the local production of functionally potent NK cells tailored for high-risk acute myeloid leukemia (AML) and neuroblastoma (NBL) patients. Moreover, the quality control strategy and considerations for product batch specifications in early clinical development are described. The protocol is based on the CliniMACS Prodigy platform and Natural Killer Cell Transduction (NKCT) (Miltenyi Biotec). NK cells are isolated from leukapheresis through CD3 depletion and CD56 enrichment, followed by a 12-hour activation with IL-2 and IL-15 cytokines. Three CliniMACS Prodigy processes demonstrated the feasibility and consistency of the modified NKCT process. A three-step process without expansion, however, compromised the NK cell yield. T cells were depleted effectively, indicating excellent safety of the product. Characterization of the NK cells before and after cytokine activation revealed a notable increase in the expression of activation markers, particularly CD69, consistent with enhanced functionality. Intriguingly, the NK cells exhibited increased killing efficacy against patient-derived CD33 + AML blasts and NBL cells in vitro, suggesting a potential therapeutic benefit in AML and NBL.
Keywords: AML blast; CliniMACS prodigy; Immunotherapy; Natural killer cells.
© 2024. The Author(s).
Conflict of interest statement
SM is an employee of Miltenyi Biotec. The authors declare no competing interests.
Figures





References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

