Molecular basis of neurodegeneration in a mouse model of Polr3-related disease
- PMID: 39499645
- PMCID: PMC11537486
- DOI: 10.7554/eLife.95314
Molecular basis of neurodegeneration in a mouse model of Polr3-related disease
Abstract
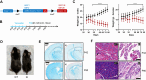
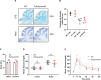
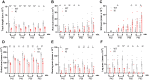
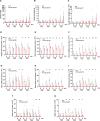
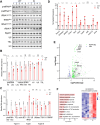
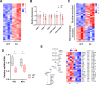
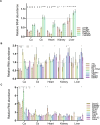
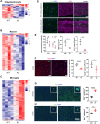
Pathogenic variants in subunits of RNA polymerase (Pol) III cause a spectrum of Polr3-related neurodegenerative diseases including 4H leukodystrophy. Disease onset occurs from infancy to early adulthood and is associated with a variable range and severity of neurological and non-neurological features. The molecular basis of Polr3-related disease pathogenesis is unknown. We developed a postnatal whole-body mouse model expressing pathogenic Polr3a mutations to examine the molecular mechanisms by which reduced Pol III transcription results primarily in central nervous system phenotypes. Polr3a mutant mice exhibit behavioral deficits, cerebral pathology and exocrine pancreatic atrophy. Transcriptome and immunohistochemistry analyses of cerebra during disease progression show a reduction in most Pol III transcripts, induction of innate immune and integrated stress responses and cell-type-specific gene expression changes reflecting neuron and oligodendrocyte loss and microglial activation. Earlier in the disease when integrated stress and innate immune responses are minimally induced, mature tRNA sequencing revealed a global reduction in tRNA levels and an altered tRNA profile but no changes in other Pol III transcripts. Thus, changes in the size and/or composition of the tRNA pool have a causal role in disease initiation. Our findings reveal different tissue- and brain region-specific sensitivities to a defect in Pol III transcription.
Keywords: Polr3-related disease; RNA polymerase III; chromosomes; gene expression; innate immune response; integrated stress response; mouse; neurodegeneration; neuroscience; tRNA.
© 2024, Moir, Merheb et al.
Conflict of interest statement
RM, EM, VC, ES, IW No competing interests declared
Figures



Update of
-
Molecular basis of neurodegeneration in a mouse model of Polr3-related disease.bioRxiv [Preprint]. 2024 Jul 2:2023.12.12.571310. doi: 10.1101/2023.12.12.571310. bioRxiv. 2024. Update in: Elife. 2024 Nov 05;13:RP95314. doi: 10.7554/eLife.95314. PMID: 38168294 Free PMC article. Updated. Preprint.
References
-
- Abbink TEM, Wisse LE, Jaku E, Thiecke MJ, Voltolini-González D, Fritsen H, Bobeldijk S, Ter Braak TJ, Polder E, Postma NL, Bugiani M, Struijs EA, Verheijen M, Straat N, van der Sluis S, Thomas AAM, Molenaar D, van der Knaap MS. Vanishing white matter: deregulated integrated stress response as therapy target. Annals of Clinical and Translational Neurology. 2019;6:1407–1422. doi: 10.1002/acn3.50826. - DOI - PMC - PubMed
-
- Anzi S, Stolovich-Rain M, Klochendler A, Fridlich O, Helman A, Paz-Sonnenfeld A, Avni-Magen N, Kaufman E, Ginzberg MB, Snider D, Ray S, Brecht M, Holmes MM, Meir K, Avivi A, Shams I, Berkowitz A, Shapiro AMJ, Glaser B, Ben-Sasson S, Kafri R, Dor Y. Postnatal exocrine pancreas growth by cellular hypertrophy correlates with a shorter lifespan in mammals. Developmental Cell. 2018;45:726–737. doi: 10.1016/j.devcel.2018.05.024. - DOI - PubMed
-
- Azmanov DN, Siira SJ, Chamova T, Kaprelyan A, Guergueltcheva V, Shearwood A-MJ, Liu G, Morar B, Rackham O, Bynevelt M, Grudkova M, Kamenov Z, Svechtarov V, Tournev I, Kalaydjieva L, Filipovska A. Transcriptome-wide effects of a POLR3A gene mutation in patients with an unusual phenotype of striatal involvement. Human Molecular Genetics. 2016;25:4302–4314. doi: 10.1093/hmg/ddw263. - DOI - PubMed
MeSH terms
Substances
Associated data
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

