Pharmacometric and statistical considerations for dose optimization
- PMID: 39501786
- PMCID: PMC11812929
- DOI: 10.1002/psp4.13271
Pharmacometric and statistical considerations for dose optimization
Abstract
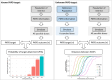
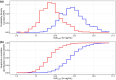
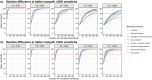
The probability of target attainment (PTA) is a common metric in drug dose optimization, but it requires a specific known target concentration threshold. Such target thresholds are not always available for some treatments, and patient and disease groups, particularly when treating children. This study performed pharmacokinetic and pharmacokinetic-pharmacodynamic (PKPD) simulations to explore different statistical approaches for determining the optimal dose for unknown PK and PKPD targets. To determine an optimal dose, PK and PKPD outcomes in typical patients with a standard adult dosing regimen were simulated and set as the reference profile, and compared to simulated outcomes for different dosing regimens in the population of interest. Statistical distances between the empirical cumulative distribution functions of the outcomes from all possible dosing regimens were calculated and compared to the reference profile. An optimal dose for known PK and PKPD target outcomes was selected to maintain the outcome above the assigned target, while optimal dosing in a population of interest with an unknown target was selected to generate equivalent PK and PKPD outcomes as the typical population. All of the dose optimization methods with commonly used PK and PKPD models and covariates were implemented as an open source freely available Shiny web-application. The developed pharmacometric method for dose optimization in populations with known and unknown target levels were robust and reproducible, and the implementation of a freely accessible Shiny web-application ensures widespread use and could be a useful tool for dose optimization in populations of interest.
© 2024 The Author(s). CPT: Pharmacometrics & Systems Pharmacology published by Wiley Periodicals LLC on behalf of American Society for Clinical Pharmacology and Therapeutics.
Conflict of interest statement
The authors declared no competing interests for this work.
Figures
Similar articles
-
Understanding and applying pharmacometric modelling and simulation in clinical practice and research.Br J Clin Pharmacol. 2017 Feb;83(2):247-254. doi: 10.1111/bcp.13119. Epub 2016 Sep 29. Br J Clin Pharmacol. 2017. PMID: 27567102 Free PMC article. Review.
-
Nedosiran population pharmacokinetic and pharmacodynamic modelling and simulation to guide clinical development and dose selection in patients with primary hyperoxaluria type 1.Br J Clin Pharmacol. 2024 Dec;90(12):3176-3189. doi: 10.1111/bcp.16194. Epub 2024 Aug 7. Br J Clin Pharmacol. 2024. PMID: 39113219 Free PMC article.
-
Pharmacokinetic/pharmacodynamic (PK/PD) indices of antibiotics predicted by a semimechanistic PKPD model: a step toward model-based dose optimization.Antimicrob Agents Chemother. 2011 Oct;55(10):4619-30. doi: 10.1128/AAC.00182-11. Epub 2011 Aug 1. Antimicrob Agents Chemother. 2011. PMID: 21807983 Free PMC article.
-
Semi-mechanistic pharmacokinetic-pharmacodynamic modelling of antibiotic drug combinations.Clin Microbiol Infect. 2018 Jul;24(7):697-706. doi: 10.1016/j.cmi.2017.11.023. Epub 2017 Dec 8. Clin Microbiol Infect. 2018. PMID: 29229429 Review.
-
gPKPDSim: a SimBiology®-based GUI application for PKPD modeling in drug development.J Pharmacokinet Pharmacodyn. 2018 Apr;45(2):259-275. doi: 10.1007/s10928-017-9562-9. Epub 2018 Jan 4. J Pharmacokinet Pharmacodyn. 2018. PMID: 29302838 Free PMC article.
References
-
- Lavielle M. mlxR: Simulation of Longitudinal Data . 2017.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

