Temperature-guided high and very high-power short duration ablation for atrial fibrillation treatment: the peQasus multicentre study
- PMID: 39504572
- PMCID: PMC12187331
- DOI: 10.1093/europace/euae284
Temperature-guided high and very high-power short duration ablation for atrial fibrillation treatment: the peQasus multicentre study
Abstract
Aims: Temperature-controlled high-power short-duration (HPSD) radiofrequency catheter ablation for pulmonary vein isolation (PVI) utilizing a novel ablation catheter (QDOT Micro) with real-time assessment of catheter tip temperature aims for safer, more effective, and faster procedures.
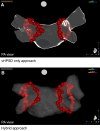
Methods and results: The peQasus study is a large European multicentre study set up to assess safety, acute efficacy, and outcomes of temperature-controlled HPSD-based PVI. The primary endpoints were safety, efficacy, and 12-month freedom from atrial tachyarrhythmias. Additionally, two strategies namely very HPSD (90 W for 4 s) only and a hybrid approach (HPSD with maximum of 50 W and vHPSD) were compared. A total of 1023 AF patients in 15 centres from nine European countries received PVI with the QDOT. Complete PVI was successfully achieved in all patients. In 699/1023 (68.3%), the vHPSD-only approach (vHPSD group) and in 324/ (31.7%) patients, the hybrid approach (hybrid group) was utilized. The mean procedure duration was 98.4 ± 37.4 min (vHPSD: 88.2 ± 34.9 min, hybrid: 117.4 ± 32.7 min, P < 0.001). The first-pass isolation rate of all PVs was 64% (vHPSD: 62.6%, hybrid: 67.1%, P = 0.187). Severe adverse events were observed in 1.7% (vHPSD: 1.6%, hybrid: 1.9%, P = 0.746). Twelve-month arrhythmia-recurrence-free survival was 77.1% (vHPSD: 76.8%, hybrid: 77.8%, P = 0.241).
Conclusion: In this large multicentre study, temperature-controlled HPSD and vHPSD ablation via a novel ablation catheter provides safe and effective PVI with a relatively short procedure duration. Despite a shorter procedure time, no differences in terms of safety and freedom from arrhythmia recurrence were found irrespective of utilizing vHPSD or the hybrid approach.
Keywords: Acute efficacy; Atrial fibrillation; High-power short duration; Pulmonary vein isolation; Radiofrequency.
© The Author(s) 2024. Published by Oxford University Press on behalf of the European Society of Cardiology.
Conflict of interest statement
Conflict of interest: C.-H.H.: received travel grants and research grants by Abbott, Boston Scientific, Biosense Webster Biotronik, Medtronic, Cardiofocus, and Lifetech and speaker’s honoraria from Abbott, Boston Scientific, Biosense Webster Biotronik, Medtronic, Cardiofocus, and Lifetech. R.R.T.: is a consultant for Boston Scientific, Philips, Medtronic Biosense Webster, and Abbott Medical, received speaker’s honoraria from Boston Scientific, Biotronik, Biosense Webster, Abbott Medical, Lifetech, and Pfizer, and research grants from Abbott, Biotronik, Medtronic, Biosense Webster, and Lifetech. He received travel grants from Abbott, Biosense Webster, Boston Scientific, Medtronic, and Philips. H.P.: consultancy and speaker fees from Abbott, Biosense Webster, Biotronik, Boston, and Medtronic. C.S.: received research support and lecture fees from Medtronic, Abbott, Boston Scientific, and Biosense Webster; is a consultant for Medtronic, Boston Scientific, and Biosense Webster; and has received grant support from the Else Kröner-Fresenius-Stiftung and Deutsche Herzstiftung. G.L.: speaker honoraria from Medtronic, Abbott, and Biosense Webster. N.S.: consulting and speaker fees from Biosense Webster, Abbott, and Boston Scientific, not related to the present study. M.R.G.: speaker bureau for Biosense Webster and Abbott. J.B.: consultancy and speaker fees from Biosense Webster, Boston, and Medtronic. All other authors: no current relevant conflicts of interest.
Figures




References
-
- Hindricks G, Potpara T, Dagres N, Arbelo E, Bax JJ, Blomström-Lundqvist C et al. 2020 ESC guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS): the task force for the diagnosis and management of atrial fibrillation of the European Society of Cardiology (ESC) developed with the special contribution of the European Heart Rhythm Association (EHRA) of the ESC. Eur Heart J 2020;42:373–498. - PubMed
-
- Kuck K-H, Brugada J, Fürnkranz A, Metzner A, Ouyang F, Chun KRJ et al. Cryoballoon or radiofrequency ablation for paroxysmal atrial fibrillation. New Engl J Medicine 2016;374:2235–45. - PubMed
-
- Hussein A, Das M, Chaturvedi V, Asfour IK, Daryanani N, Morgan M et al. Prospective use of ablation index targets improves clinical outcomes following ablation for atrial fibrillation. J Cardiovasc Electrophysiol 2017;28:1037–47. - PubMed
-
- Hussein A, Das M, Riva S, Morgan M, Ronayne C, Sahni A et al. Use of ablation index-guided ablation results in high rates of durable pulmonary vein isolation and freedom from arrhythmia in persistent atrial fibrillation patients. Circulation Arrhythmia Electrophysiol 2018;11:e006576. - PubMed
-
- Kottmaier M, Popa M, Bourier F, Reents T, Cifuentes J, Semmler V et al. Safety and outcome of very high-power short-duration ablation using 70 W for pulmonary vein isolation in patients with paroxysmal atrial fibrillation. Europace 2019;22:388–93. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Research Materials

