Differences in immune-related toxicity between PD-1 and PD-L1 inhibitors: a retrospective cohort study in patients with advanced cancer
- PMID: 39508883
- PMCID: PMC11543953
- DOI: 10.1007/s00262-024-03869-1
Differences in immune-related toxicity between PD-1 and PD-L1 inhibitors: a retrospective cohort study in patients with advanced cancer
Abstract
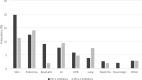
Immunotherapy with PD-1 or PD-L1 inhibitors has become an essential treatment strategy for a growing number of malignancies. These treatments have a risk for immune-related adverse events (IRAEs). Pooled analyses based on clinical trials show a favorable toxicity profile for treatment with PD-L1 compared to PD-1 inhibitors. This study aimed to investigate differences in IRAEs between patients with advanced solid malignances treated with PD-L1 and PD-1 inhibitors in a real-world setting. We conducted a retrospective cohort study at four Swedish Regions. Patients (n = 605) treated for advanced cancer with a PD-L1 or PD-1 inhibitor in monotherapy between June 2016 and August 2022 were included. Non-small cell lung cancer (NSCLC) was the most common malignant disease (n = 251; 41.5%), followed by malignant melanoma (n = 173; 28.6%), renal cell carcinoma (n = 71; 11.7%) and urothelial carcinoma (n = 35; 5.8%). Among patients receiving PD-L1 inhibitors, NSCLC (94.4%) was the predominant malignancy, whereas for those treated with PD-1 inhibitor, malignant melanoma constituted the most prevalent malignancy (34.5%). Discontinuation of treatment due to IRAEs overall and IRAEs grade ≥ 2 were significantly less common in patients treated with PD-L1 compared to PD-1 inhibitors [Odds Ratio (OR): 0.38 (95% Confidence Interval (CI) 0.16-0.88) and OR: 0.63 (95% CI 0.35-0.98) respectively]. Any grade IRAE, IRAE grade ≥ 3 and multiple IRAEs were numerically more frequent in patients treated with PD-1 inhibitors.In conclusion, our study of patients with advanced solid malignancies in a real-world setting supports the results from clinical trials demonstrating a favorable toxicity profile for PD-L1 inhibitors versus PD-1 inhibitors.
Keywords: Advanced cancer; Cohort study; Immune-related adverse events; PD-1 inhibitors; PD-L1 inhibitors.
© 2024. The Author(s).
Conflict of interest statement
No potential conflict of interest was reported by the authors.
Figures
References
-
- CRI (2023) Regulatory Approval Timeline of Active immunotherapies 20240109]; Available from: https://www.cancerresearch.org/regulatory-approval-timeline-of-active-im...
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials