Primary Open-Angle Glaucoma Polygenic Risk Score and Risk of Disease Onset: A Post Hoc Analysis of a Randomized Clinical Trial
- PMID: 39509108
- PMCID: PMC11544551
- DOI: 10.1001/jamaophthalmol.2024.4376
Primary Open-Angle Glaucoma Polygenic Risk Score and Risk of Disease Onset: A Post Hoc Analysis of a Randomized Clinical Trial
Abstract
Importance: Primary open-angle glaucoma (POAG) is a heritable disease. A polygenic risk score (PRS) threshold may be used to identify individuals at low risk of disease onset.
Objective: To assess the utility of a POAG PRS to identify ocular hypertensive individuals at low risk of disease onset.
Design, setting, and participants: This is a post hoc analysis of the Ocular Hypertension Treatment Study (OHTS), a multicenter randomized clinical trial across 22 centers in the US conducted among 1636 participants with ocular hypertension from February 1994 to April 2019 with available genetic data. Of the 1636 original participants, 1077 had available genetic data; after excluding 67 for missing data, data quality concerns, or ancestry other than European or African, 1010 were included in the present analysis. Data for this report were analyzed from November 2023 to June 2024.
Exposure: From 1994-2002, participants were randomized to receive topical intraocular pressure (IOP)-lowering medications. From 2002 onwards, all participants were given topical IOP-lowering medications.
Main outcome and measure: Twenty-year conversion rates by POAG PRS threshold, baseline randomization status, and OHTS clinical risk tertile.
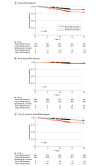
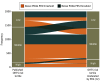
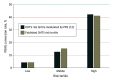
Results: Among the 1010 participants in this study, 563 (65.8%) were female, and the mean (SD) age was 55.9 (9.4) years. In a mixed-effects logistic regression model adjusted for OHTS risk factors for conversion to POAG and randomization status, a PRS under the 48th percentile was associated with a 1.49 times higher likelihood of disease-free status after 20 years of follow-up (95% CI, 1.04-2.15; P = .03; unadjusted hazard ratio [HR], 1.64; 95% CI, 1.13-2.38; P = .009), compared with high polygenic risk. When we stratified the trial cohort into nongenetic OHTS clinical risk tertiles, the largest differences in survival probability at 20 years based on PRS threshold was observed in eyes in the highest tertile, initial observation group (20-year conversion rate: 61.1% in the high polygenic risk group vs 23.8% in the low polygenic risk group; 95% CI, -63.0 to -11.6; P = .01), with randomization to early treatment partially mitigating the effect of high genetic risk (20-year conversion rate: 37.3% in the high polygenic risk group vs 24.1% in the low polygenic risk group; 95% CI, -35.6 to 9.3%; P = .32).
Conclusions and relevance: These findings support considering use of a POAG PRS threshold to identify individuals at low risk of disease onset, with those below the PRS threshold more likely to have lower conversion rates over 20 years. Among those considered at highest risk based on the OHTS clinical risk model, early treatment may partially offset the association with high genetic risk but provide limited benefit for those with low genetic risk.
Trial registration: ClinicalTrials.gov Identifier: NCT00000125.
Conflict of interest statement
Figures

Comment in
References
-
- Kass MA, Heuer DK, Higginbotham EJ, et al. The Ocular Hypertension Treatment Study: a randomized trial determines that topical ocular hypotensive medication delays or prevents the onset of primary open-angle glaucoma. Arch Ophthalmol. 2002;120(6):701-713. doi: 10.1001/archopht.120.6.701 - DOI - PubMed
-
- Kass MA, Heuer DK, Higginbotham EJ, et al. ; Ocular Hypertension Study Group . Assessment of cumulative incidence and severity of primary open-angle glaucoma among participants in the ocular hypertension treatment study after 20 years of follow-up. JAMA Ophthalmol. 2021;139(5):1-9. doi: 10.1001/jamaophthalmol.2021.0341 - DOI - PMC - PubMed
Associated data
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

