Prospective observational study and mechanistic evidence showing lipolysis of circulating triglycerides worsens hypertriglyceridemic acute pancreatitis
- PMID: 39509346
- PMCID: PMC11684810
- DOI: 10.1172/JCI184785
Prospective observational study and mechanistic evidence showing lipolysis of circulating triglycerides worsens hypertriglyceridemic acute pancreatitis
Abstract
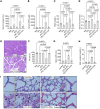
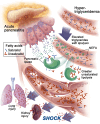
BACKGROUNDWhile most hypertriglyceridemia is asymptomatic, hypertriglyceridemia-associated acute pancreatitis (HTG-AP) can be more severe than AP of other etiologies. The reasons underlying this are unclear. We thus examined whether lipolytic generation of nonesterified fatty acids (NEFAs) from circulating triglycerides (TGs) could worsen clinical outcomes.METHODSAdmission serum TGs, NEFA composition, and concentrations were analyzed prospectively for 269 patients with AP. These parameters, demographics, and clinical outcomes were compared between HTG-AP (TGs >500 mg/dL; American Heart Association [AHA] 2018 guidelines) and AP of other etiologies. Serum NEFAs were correlated with serum TG fatty acids (TGFAs) alone and with the product of TGFA serum lipase (NEFAs - TGFAs × lipase). Studies in mice and rats were conducted to understand the role of HTG lipolysis in organ failure and to interpret the NEFA-TGFA correlations.RESULTSPatients with HTG-AP had higher serum NEFA and TG levels and more severe AP (19% vs. 7%; P < 0.03) than did individuals with AP of other etiologies. Correlations of long-chain unsaturated NEFAs with corresponding TGFAs increased with TG concentrations up to 500 mg/dL and declined thereafter. However, NEFA - TGFA × lipase correlations became stronger with TGs above 500 mg/dL. AP and intravenous lipase infusion in rodents caused lipolysis of circulating TGs to NEFAs. This led to multisystem organ failure, which was prevented by pancreatic TG lipase deletion or lipase inhibition.CONCLUSIONSHTG-AP is made severe by the NEFAs generated from intravascular lipolysis of circulating TGs. Strategies that prevent TG lipolysis may be effective in improving clinical outcomes for patients with HTG-AP.FUNDINGNational Institute of Diabetes and Digestive and Kidney Diseases (NIDDK, NIH) (RO1DK092460 and R01DK119646); Department of Defense (PR191945 under W81XWH-20-1-0400); National Institute on Alcohol Abuse and Alcoholism (NIAAA), NIH (R01AA031257).
Keywords: Gastroenterology; Lipoproteins.
Conflict of interest statement
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

