Cell Membrane-Coated Nanotherapeutics for the Targeted Treatment of Acute and Chronic Colitis
- PMID: 39512421
- PMCID: PMC11542430
- DOI: 10.34133/bmr.0102
Cell Membrane-Coated Nanotherapeutics for the Targeted Treatment of Acute and Chronic Colitis
Abstract
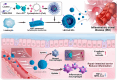
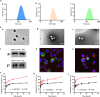
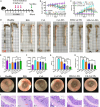
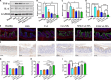
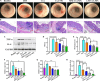
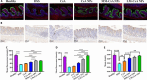
Integrin α4β1 and α4β7 are overexpressed in macrophages and leukocytes and play important roles in mediating cell homing and recruitment to inflammatory tissues. Herein, to enhance the targeting ability of nanotherapeutics for inflammatory bowel disease (IBD) treatment, cyclosporine A-loaded nanoparticles (CsA NPs) were coated with macrophage membranes (MM-CsA NPs) or leukocyte membranes (LM-CsA NPs). In vitro experiments demonstrated that the physicochemical properties of the nanotherapeutics (e.g., size, zeta potential, polymer dispersity index, and drug release profiles) did not obviously change after cell membrane coating. However, integrin α4β1 and α4β7 were expressed in MM-CsA NPs and LM-CsA NPs, respectively, which significantly inhibited normal macrophage phagocytosis and obviously increased uptake by proinflammatory macrophages and endothelial cells. In vivo experiments verified that cell membrane-coated nanotherapeutics have longer retention times in inflammatory intestinal tissues. Importantly, LM-CsA NPs significantly mitigated weight loss, alleviated colon shortening, decreased disease activity indices (DAIs), and promoted colon tissue repair in acute and chronic colitis model mice. Furthermore, LM-CsA NPs significantly decreased the expression of inflammatory factors such as TNF-α and IL-6 and increased the expression of gut barrier-related proteins such as E-cadherin, ZO-1, and occludin protein in colitis mice.
Copyright © 2024 Shan Li et al.
Conflict of interest statement
Competing interests: The authors declare that they have no competing interests.
Figures


Similar articles
-
Colon-targeted delivery of cyclosporine A using dual-functional Eudragit® FS30D/PLGA nanoparticles ameliorates murine experimental colitis.Int J Nanomedicine. 2018 Feb 28;13:1225-1240. doi: 10.2147/IJN.S157566. eCollection 2018. Int J Nanomedicine. 2018. PMID: 29535519 Free PMC article.
-
Cyclosporine A-Encapsulated pH/ROS Dual-Responsive Nanoformulations for the Targeted Treatment of Colitis in Mice.ACS Biomater Sci Eng. 2023 Oct 9;9(10):5737-5746. doi: 10.1021/acsbiomaterials.3c01191. Epub 2023 Sep 21. ACS Biomater Sci Eng. 2023. PMID: 37733924
-
Enhanced therapeutic efficacy of a novel colon-specific nanosystem loading emodin on DSS-induced experimental colitis.Phytomedicine. 2020 Nov;78:153293. doi: 10.1016/j.phymed.2020.153293. Epub 2020 Jul 25. Phytomedicine. 2020. PMID: 32777486
-
Surface modification potentials of cell membrane-based materials for targeted therapies: a chemotherapy-focused review.Nanomedicine (Lond). 2023 Aug;18(19):1281-1303. doi: 10.2217/nnm-2023-0164. Epub 2023 Sep 27. Nanomedicine (Lond). 2023. PMID: 37753724 Review.
-
Development, validation and implementation of an in vitro model for the study of metabolic and immune function in normal and inflamed human colonic epithelium.Dan Med J. 2015 Jan;62(1):B4973. Dan Med J. 2015. PMID: 25557335 Review.
Cited by
-
Nucleic Acid-Functionalized Gold Nanorods Modulate Inflammation and Dysregulated Intestinal Barriers for Treatment of Ulcerative Colitis.Biomater Res. 2025 Apr 18;29:0195. doi: 10.34133/bmr.0195. eCollection 2025. Biomater Res. 2025. PMID: 40255504 Free PMC article.
References
-
- Kuenzig ME, Fung SG, Marderfeld L, Mak JWY, Kaplan GG, Ng SC, Wilson DC, Cameron F, Henderson P, Kotze PG, et al. . Twenty-first century trends in the global epidemiology of pediatric-onset inflammatory bowel disease: Systematic review. Gastroenterology. 2022;162(4):1147–1159.e4. - PubMed
-
- Hoivik ML, Moum B, Solberg IC, Henriksen M, Cvancarova M, Bernklev T; IBSEN Group. Work disability in inflammatory bowel disease patients 10 years after disease onset: Results from the IBSEN Study. Gut. 2013;62(3):368–375. - PubMed
LinkOut - more resources
Full Text Sources

