Sitravatinib in patients with solid tumors selected by molecular alterations: results from a Phase Ib study
- PMID: 39513224
- PMCID: PMC11654803
- DOI: 10.1080/14796694.2024.2418285
Sitravatinib in patients with solid tumors selected by molecular alterations: results from a Phase Ib study
Abstract
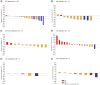
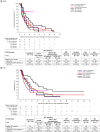
Aim: We report clinical activity and safety of sitravatinib in patients with advanced cancer from basket cohorts with specific molecular alterations, in a Phase Ib study.Materials & methods: Patients with advanced solid tumors harboring amplification, mutation, or rearrangement of MET, AXL, RET, NTRK, DDR2, KDR, PDGFRA, KIT or CBL received sitravatinib once daily. Primary end point was confirmed objective response rate (ORR).Results: In total, 113 patients were enrolled following a median of 3 (range 1-18) prior systemic regimens. Altered RET (n = 31), CBL (n = 31) and MET (n = 17) were most frequent cohorts. Overall, 68.9% had reduced tumor volume and most (61.5%) had a best objective response of stable disease. ORR was highest in patients with RET-rearranged non-small cell lung cancer (21.1%) but did not differ significantly from the null hypothesis (ORR ≤15%; p = 0.316). Median progression-free survival and overall survival (5.7 and 24.2 months, respectively) were also longest in the RET-rearranged non-small cell lung cancer cohort. Diarrhea (61.1%), fatigue (50.4%) and hypertension (46.9%) were the most frequent treatment-emergent adverse events. Most treatment-emergent adverse events were mild-to-moderate in severity. The study closed before the planned number of patients were enrolled in all cohorts.Conclusion: Sitravatinib had a manageable safety profile with modest signals of clinical activity in patients with molecularly selected solid tumors.Clinical trial registration: www.clinicaltrials.gov identifier is NCT02219711.
Keywords: MGCD516; basket study; molecular alteration; sitravatinib; solid tumor.
Plain language summary
We report findings from a clinical study of sitravatinib which included patients with cancer that could not be removed by surgery or had spread to other parts of the body. The tumors of these patients contained specific molecular changes in one of the following genes: MET, AXL, RET, NTRK, DDR2, KDR, PDGFRA, KIT or CBL. All patients received treatment with sitravatinib once a day. Change in tumor size over time was assessed to see how effective treatment with sitravatinib was.In total, 113 patients joined the study. Most patients had already received a median of three different types of medicines for their cancer (and up to 18 different types of anticancer medicines). Most patients had tumors that contained alterations in RET, CBL or MET genes.During the study, the percentage of patients who had a decrease in the tumor size was highest in the group with non-small cell lung cancer that contained an altered RET gene (21.1%). However, this level of response to sitravatinib was not considered high enough to be medically important.The most common side effects during the study were diarrhea (61.1%), fatigue (50.4%) and high blood pressure (46.9%). Most side effects were mild or moderate in severity. The study provided the opportunity to assess sitravatinib as a treatment for cancers with specific gene mutations that are uncommon; the study closed before the planned number of patients were enrolled.In conclusion, the side effects seen in patients who received sitravatinib were manageable. Signals of how well sitravatinib worked were modest in patients with cancer that had spread to other parts of the body and contained specific molecular changes.
Conflict of interest statement
L Bazhenova: consulting fees from BioAtla, Abbvie, Gilead, Anheart, Bayer, Boehringer Ingelheim, Janssen, Bristol Myers Squibb, Daichi Sankyo, Sanofi,Elevation Oncology, Genentech, Intervenn, Merck, Neuvogen, Novocure, Pfizer, Regeneron, Summit Therapeutics, Teligene, Mirati Therapeutics, and Turning Point Therapeutics; consulting fees paid to institution from AstraZeneca and Takeda; Data Safety Monitoring Board/Advisory Board participation for Neuvogen and ORIC Pharmaceuticals; and stock/stock options for Epic Sciences. D-W Kim: research funding to institution from Alpha Biopharma, BridgeBioTherapeutics, Amgen, AstraZeneca/Medimmune, Boehringer-Ingelheim, GSK, Bristol Myers Squibb, TP Therapeutics, Chong Keun Dang, Novartis, Daiichi-Sankyo, Hanmi, IMBDx, InnoN, Janssen, Merck, Meurs, Mirati Therapeutics, MSD, ONO Pharmaceutical, Pfizer, Roche/Genentech, Takeda, Xcovery, and Yuhan; honoraria from Korean Association for Lung Cancer, Asian Thoracic Oncology Research Group, Korean Cancer Association, Korean Society of Medical Oncology, and Taiwan Lung Cancer Society; support for meeting attendance from International Association for the Study of Lung Cancer, Asian Thoracic Oncology Research Group, and Taiwan Lung Cancer Society; advisory board participation (unpaid) from Bristol Myers Squibb/ONO Pharmaceuticals, Amgen, Merck, AstraZeneca, Daiichi-Sankyo, GSK, Novelty Nobility, Janssen, MSD, Oncobix, Pfizer, SK Biopharm, and Takeda; scientific advisor for Health Insurance Review and Assessment Service, Republic of Korea; and member of Board of Directors for Korean Cancer Association, Asian Thoracic Oncology Research Group, Korean Association for Lung Cancer, and Korean Society of Medical Oncology. BC Cho: research funding from Abbvie, Dizal Pharma, Abion, Bayer, AstraZeneca, Blueprint Medicines, Boehringer Ingelheim, CJ Bioscience, Bridgebio Therapeutics, CHA Bundang Medical Center, JINTSbio, Champions Oncology, CJ Blossom Park, Lilly, Cyrus, MOGAM Institute, Dong-A ST, Genexine, GI-Cell, Illumina, GI-ImmuneOncia, Kanaph Therapeutics, Hanmi, Innovation, Interpark Bio Convergence Corp, Regeneron, Janssen, LG Chem, MSD, Novartis Oncternal, Ono, Oscotec, Therapex, Nuvalent, Vertical Bio AG, and Yuhan; consulting fees from BeiGene, Abion, Pfizer, Cyrus Therapeutics, AstraZeneca, Hanmi, Blueprint Medicines, Eli Lilly, Boehringer-Ingelheim, Onegene Biotechnology, CJ, CureLogen, GI-Cell, Guardant, HK Inno-N, Imnewrun Biosciences Inc., Bristol Myers Squibb, Janssen, Ono, Medpacto, MSD, Novartis, RandBio, Takeda, and Yuhan; honoraria from Chinese Thoracic Oncology Society, American Society of Clinical Oncology, International Association for the Study of Lung Cancer, AstraZeneca, Korean Cancer Association, European Society of Medical Oncology, Guardant, Korean Cancer Study Group, Korean Society of Medical Oncology; MSD, Novartis, Korean Society of Thyroid, Head and Neck Surgery, Roche, and Pfizer; advisory board roles for Guardant Health, Amgen, Cyrus Therapeutics, Bridgebio Therapeutics, KANAPH Therapeutic Inc, Gilead, J INTS BIO, Oscotec Inc, and Therapex; royalties from Crown Bioscience, Bio GmbH, Champions Oncology, Imagen, PearlRiver, and Roche; member of the board of directors for J INTS BIO; founder of DAAN Biotherapeutics; and stock ownership for Cyrus Therapeutics, Gencurix Inc, Bridgebio Therapeutics, Interpark Bio Convergence Corp., J INTS BIO, KANAPH Therapeutics, and TheraCanVac Inc. S Goel: research support to institution from Mirati Therapeutics Inc. R Heist: research funding to institution from Daiichi Sankyo, Abbvie, Corvus, Erasca, Lilly, Mirati Therapeutics, Agios, Mythic, Novartis, and Turning Point; and consulting fees from Abbvie, Claim Therapeutics, Amgen, Regeneron, EMD Serono, AstraZeneca, Daiichi Sankyo, Lilly, Merck, Novartis, and Sanofi. TL Werner: research support to institution from Abbvie, Mersana Therapeutics, Acrivon, AstraZeneca, Genmab, BluePrint Medicines, GSK-Tesaro, Repare Therapeutics, Clovis Oncology, and Roche Genetech; and honoraria for advisory roles from Mersana Therapeutics. KD Eaton: research support to institution from Mirati Therapeutics Inc. JS Wang: consulting fees from Kanaph Therapeutics, honoraria from AstraZeneca and Eisai; and advisory roles for Janssen Oncology, BioNTech, Fusion Pharmaceuticals, and Stemline/Menarini. S Pant: research support to institution for Astellas, Amal Therapeutics, Bristol Myers Squibb, Arcus, Lilly, Biontech, 4D Pharma, Elicio, Framewave, Immuneering, ImmunoMET, Ipsen, Janssen, Boehringer Ingelheim, Mirati Therapeutics, NGM Pharmaceuticals, Pfizer, Xencor, Novartis, and, Zymeworks; and consulting/advisory fees from AstraZeneca, Jazz Pharmaceuticals, Alligator Bioscience, AskGene Pharma, BPGBio, Ipsen, Janssen, Boehringer Ingelheim, Nihon Medi-Physics Co, Ltd, USWorldmeds, Novartis, and Zymeworks. DR Adkins: research support to institution from Beigene, Adlai Nortye, Pfizer, Calliditas, AstraZeneca, BioAtla, Celgene/Bristol Meyers Squibb, Cue Biopharma, Gilead Sciences, Debiopharm, Boehringer Ingelheim, Eli Lilly, Epizyme, Blueprint Medicine, Genmab, Cofactor Genomics, Hookipa, ISA Pharmaceuticals, Kura Oncology, Immutep, Merck, Mirati Therapeutics, Novartis, Roche, Tizona, Natco, and Vaccinex; consulting fees for Advisory Board participation from Coherus Biosciences, Blueprint Medicine, Gilead Sciences, Boehringer Ingelheim, Cue Biopharma, Immunitas, Eisai Europe, Exelixis, Jazz Pharmaceuticals, Merck, Kura Oncology, Targimmue Therapeutics, Vaxxinex, TwoXAR, and Xilio Therapeutics; travel support for meeting attendance to institution from Natco; Chairperson for Washington University Standing Data Safety Monitoring Board; Chairperson of BJC Pharmacy and Therapeutics Committee; and member of NCI Head and Neck Cancer Steering Committee. CM Blakely: research support from AstraZeneca, Puma, Mirati Therapeutics Inc., Genentech, Novartis, Pfizer, and Takeda; and consulting fees from Bayer, Gilead, Bristol Meyers Squibb, and Janssen. X Yan: employment, stock ownership and support for meeting attendance for Mirati Therapeutics Inc. S Neuteboom: prior employment and stock ownership for Mirati Therapeutics Inc. JG Christensen: employment and stock ownership for Mirati Therapeutics Inc., and Fiduciary Officer for Mirati Therapeutics Inc. and Boundless Bio. R Chao: employment, stock ownership and support for meeting attendance for Mirati Therapeutics Inc. T Bauer: consulting fees from Pfizer, Bayer, Lilly, and Sanofi; honoraria from Lilly, Bayer, and Pfizer; and participation on an Advisory Board/Data Safety Monitoring Board for Pfizer. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
Figures

References
-
- Bauer T, Cho BC, Heist R, et al. . First-in-human Phase I/Ib study to evaluate sitravatinib in patients with advanced solid tumors. Invest New Drugs. 2022;40(5):990–1000. doi:10.1007/s10637-022-01274-y - DOI - PMC - PubMed
-
•• Describes the design of this study and pharmacokinetic/pharmacodynamic, safety and efficacy findings at primary data cutoff.
-
- Patwardhan PP, Ivy KS, Musi E, et al. . Significant blockade of multiple receptor tyrosine kinases by MGCD516 (sitravatinib), a novel small molecule inhibitor, shows potent anti-tumor activity in preclinical models of sarcoma. Oncotarget. 2016;7(4):4093–5109. doi:10.18632/oncotarget.6547 - DOI - PMC - PubMed
-
•• Demonstrates potent antitumor activity of sitravatinib in sarcoma xenograft models.
Publication types
MeSH terms
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous
