Real-World and Patient-Reported Outcomes of Dupilumab and Other Biological Drugs for Chronic Obstructive Pulmonary Disease-A Systematic Review
- PMID: 39518358
- PMCID: PMC11545442
- DOI: 10.3390/diagnostics14212390
Real-World and Patient-Reported Outcomes of Dupilumab and Other Biological Drugs for Chronic Obstructive Pulmonary Disease-A Systematic Review
Abstract
Background: Over the last few decades, the efficacy of biological therapies for COPD has been evaluated by different randomized controlled trials (RCTs). Still, the evaluation of real-world data and patient-reported outcome measures (PROMs) have not been performed in this field before. In the current work, we present a systematic literature review of the real-world data and PROMs of biological treatments for COPD.
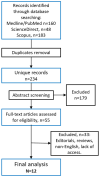
Methods: Three large databases (MEDLINE/PubMed, Scopus, and ScienceDirect) were utilized for the systematic literature review. Clinical studies (RCT, cohorts, case series/reports) assessing patients with COPD treated by any biological therapy were included.
Results: The review resulted in twelve eligible studies (nine randomized controlled trials and three "real-world" case series/reports). The evaluation of PROMs in the included studies was mainly limited to the severity and burden of respiratory symptoms. Most biological therapies were associated with improved PROMs compared to the baseline, although not for the placebo. Dupilumab was the only biologic therapy with proven efficacy in RCT for both objective and subjective measures. One prior study reported patients' self-perceived drug effects, and none evaluated patients' perceived disease status. Only 25 patients were assessed in a real-world setting for all biologic therapies combined. Real-world data were retrospective in the form of case reports or series.
Conclusions: There are limited data on patients' experience with biological therapies for COPD. While real-world data and PROMs are missing, biases such as a placebo effect must be considered, requiring their incorporation with objective outcomes from prospective controlled trials.
Keywords: biologic therapy; chronic obstructive pulmonary disease; eosinophils; exacerbations; immunologic treatment.
Conflict of interest statement
A.B.S reports receiving personal consulting fees and lecture fees from Sanofi-Regeneron, Astrazeneca, GSK, Kamada, Boehringer Ingelheim, Roche. All other authors report no conflicts of interest.
References
-
- Agustí A., Celli B.R., Criner G.J., Halpin D., Anzueto A., Barnes P., Bourbeau J., Han M.K., Martinez F.J., Montes de Oca M., et al. Global Initiative for Chronic Obstructive Lung Disease 2023 Report: GOLD Executive Summary. Am. J. Respir. Crit. Care Med. 2023;207:819–837. doi: 10.1164/rccm.202301-0106PP. - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources