1-Piperidine Propionic Acid Protects from Septic Shock Through Protease Receptor 2 Inhibition
- PMID: 39519216
- PMCID: PMC11547144
- DOI: 10.3390/ijms252111662
1-Piperidine Propionic Acid Protects from Septic Shock Through Protease Receptor 2 Inhibition
Abstract
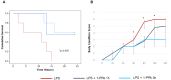
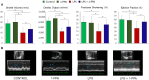
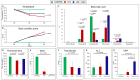
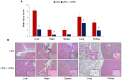
Sepsis is a complex disorder caused by a dysregulated host response to infection, with high levels of morbidity and mortality. Treatment aimed to modulate immune response and maintain vascular function is still one of the major clinical challenges. This study was designed to test the effect of the small molecule 1-Piperidine Propionic Acid (1-PPA) as molecular targeted agent to block protease-activated receptor 2 (PAR2), one of the major modulators of inflammatory response in LPS-induced experimental endotoxemia. In the THP-1 cell line, LPS-induced cytokine expression was inhibited by 1-PPA in a dose-dependent manner. In LPS-injected mice, treatment with 1-PPA was effective in reducing mortality and sepsis-related symptoms and improved cardiac function parameters. After 6 h from LPS injection, a significant decrease in IL-6, IL-1β, and IL-10 was observed in the lung tissue of 1-PPA-treated mice, compared to controls. In these mice, a significant decrease in vasoactive molecules, especially kininogen-1, was also observed, mainly in the liver. Histopathological analysis confirmed typical features of sepsis in different organs and these findings were markedly reduced in mice treated with 1-PPA. These data demonstrate the effectiveness of 1-PPA in protecting the whole organism from sepsis-induced damage.
Keywords: 1-piperidine propionic acid; experimental endotoxemia; protease-activated receptor 2; septic shock.
Conflict of interest statement
Biasiolo, A., Chinellato, M., Quarta, S., Pontisso, P., Ruvoletto, M., and Villano, G. are inventors of the Patent Application of the University of Padova N. 102022000014593. No conflicts of interest exist for the other authors.
Figures



Similar articles
-
Synergistic therapeutic potential of dexamethasone and L-arginine in lipopolysaccharide-induced septic shock.J Surg Res. 2007 Jun 1;140(1):99-108. doi: 10.1016/j.jss.2006.09.002. Epub 2007 Feb 8. J Surg Res. 2007. PMID: 17292408
-
Effects of protease activated receptor (PAR)2 blocking peptide on endothelin-1 levels in kidney tissues in endotoxemic rat mode.Life Sci. 2014 May 2;102(2):127-33. doi: 10.1016/j.lfs.2014.03.013. Epub 2014 Mar 16. Life Sci. 2014. PMID: 24641950
-
Effects of Thrombomodulin in Reducing Lethality and Suppressing Neutrophil Extracellular Trap Formation in the Lungs and Liver in a Lipopolysaccharide-Induced Murine Septic Shock Model.Int J Mol Sci. 2021 May 6;22(9):4933. doi: 10.3390/ijms22094933. Int J Mol Sci. 2021. PMID: 34066510 Free PMC article.
-
Interleukin-30 (IL27p28) alleviates experimental sepsis by modulating cytokine profile in NKT cells.J Hepatol. 2016 May;64(5):1128-1136. doi: 10.1016/j.jhep.2015.12.020. Epub 2016 Jan 6. J Hepatol. 2016. PMID: 26767500 Free PMC article.
-
[Protective effects of vasoactive intestinal peptide on intestinal lesions induced by endotoxic shock in rat].Zhonghua Er Ke Za Zhi. 2006 May;44(5):369-73. Zhonghua Er Ke Za Zhi. 2006. PMID: 16780716 Chinese.
Cited by
-
The Role of PAR2 in MASLD Progression and HCC Development.Int J Mol Sci. 2025 Jul 23;26(15):7076. doi: 10.3390/ijms26157076. Int J Mol Sci. 2025. PMID: 40806209 Free PMC article. Review.
References
-
- Rudd K.E., Johnson S.C., Agesa K.M., Shackelford K.A., Tsoi D., Kievlan D.R., Colombara D.V., Ikuta K.S., Kissoon N., Finfer S., et al. Global, regional, and national sepsis incidence and mortality, 1990–2017: Analysis for the Global Burden of Disease Study. Lancet. 2020;395:211–2020. doi: 10.1016/S0140-6736(19)32989-7. - DOI - PMC - PubMed
-
- Rhodes A., Evans L.E., Alhazzani W., Levy M.M., Antonelli M., Ferrer R., Kumar A., Sevransky J.E., Sprung C.L., Nunnally M.E., et al. Surviving sepsis campaign: International guidelines for management of sepsis and septic shock: 2016. Intensive Care Med. 2017;43:377–2017. doi: 10.1007/s00134-017-4683-6. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

