Response of One-Carbon Biomarkers in Maternal and Cord Blood to Folic Acid Dose During Pregnancy
- PMID: 39519534
- PMCID: PMC11547940
- DOI: 10.3390/nu16213703
Response of One-Carbon Biomarkers in Maternal and Cord Blood to Folic Acid Dose During Pregnancy
Abstract
Background/objectives: The folate Recommended Daily Allowance (RDA) for pregnant women is 600 μg/day dietary folate equivalents, which is equivalent to approximately 400 μg folic acid. Many prenatal supplements contain much higher doses of folic acid. The body's ability to reduce synthetic folic acid to the metabolically active form may be exceeded with high levels of supplementation. The objective of this double-blinded randomized controlled intervention trial was to determine changes in unmetabolized folic acid and other biomarkers of folate and one-carbon metabolism in maternal and cord blood in response to a folic acid dose commonly found in prenatal supplements (800 μg/day) compared to the dose equivalent of the RDA (400 μg/day).
Methods: Healthy pregnant women were randomized and provided supplements from their first prenatal visit (<12 weeks gestation) through delivery. Maternal blood was collected at baseline and delivery. Umbilical cord blood from the mothers was collected at delivery.
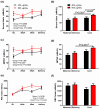
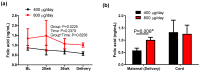
Results: A repeated measures analysis of variance revealed that there was a significant group supplemental dose effect (p = 0.0225) on serum unmetabolized folic acid concentration in mothers but no difference in cord blood unmetabolized folic acid concentrations between groups. Mixed effects analysis found a significant overall effect of pre-pregnancy BMI (p = 0.0360) and length of previous folic acid supplementation (p = 0.0281) on serum folate concentrations. No treatment effect was seen in RBC folate concentrations. Choline concentrations were higher in cord blood from the 800 μg/day group compared to the 400 μg/day group, but there was no group effect in maternal choline concentrations.
Conclusions: The results indicate that folic acid dose during pregnancy affects certain folate and one-carbon biomarkers, and these effects are not consistent between maternal and cord blood. Potential long-term effects of these results on both mothers and offspring are unknown and merit further investigation.
Keywords: folate; supplementation; unmetabolized folic acid.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


References
-
- Stover P.J. Folate biochemical pathways and their regulation. In: Bailey L., editor. Folate in Health and Disease. 2nd ed. CRC Press; Boca Raton, FL, USA: 2010. pp. 50–70.
-
- Cunningham F.G., MacDonald P.C., Gant N.F., Leveno K.G., Gilstrap L.C. Williams Obstetrics. Appleton & Lange; Norwalk, CT, USA: 1989. Physiology in pregnancy; pp. 209–247.
-
- Institute of Medicine . Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline. National Academies Press (US) National Academy of Sciences; Washington, DC, USA: 1998. The National Academies Collection: Reports funded by National Institutes of Health. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

