Management Patterns of Teduglutide Use in Short Bowel Syndrome: A Survey of 70 Healthcare Professionals
- PMID: 39519595
- PMCID: PMC11547446
- DOI: 10.3390/nu16213762
Management Patterns of Teduglutide Use in Short Bowel Syndrome: A Survey of 70 Healthcare Professionals
Abstract
Background: This study aimed to gain real-world insights from healthcare professionals (HCPs) regarding the management of adult patients with short bowel syndrome and intestinal failure (SBS-IF) who received teduglutide and achieved parenteral support (PS) independence or PS volume stability for ≥12 months.
Methods: This cross-sectional survey was conducted in five European countries and Canada via a self-reported questionnaire (November 2022-March 2023) among HCPs who manage patients with SBS-IF and who had prescribed teduglutide to ≥5 patients with SBS-IF receiving PS.
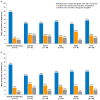
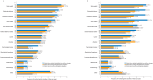
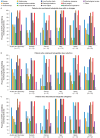
Results: Of the 70 HCPs who completed the survey, almost all reported managing patients with SBS-IF who achieved PS independence or PS volume stability (99%, 69/70 and 97%, 68/70, respectively) and maintained the standard teduglutide dose, without changes. A total of 52 HCPs managed patients who achieved PS independence and discontinued teduglutide. Of these HCPs, 73% (38/52) anticipated that these patients would remain PS-independent, not requiring PS reintroduction. Of the remainder, 79% (11/14) estimated that ≤40% of these patients would require PS reintroduction. While many HCPs (81%, 42/52) would reintroduce teduglutide in patients who discontinued its use after achieving PS independence, none would do so for patients who discontinued teduglutide after achieving PS volume stability if a patient's condition worsened.
Conclusions: This survey found that patients with SBS-IF can achieve PS independence or PS volume stability with teduglutide treatment. However, some HCPs (27%, 14/52) believe that a proportion of patients discontinuing teduglutide after achieving PS independence will require PS reintroduction. This survey suggests that teduglutide treatment should continue uninterrupted, unless clinically indicated, but this requires confirmation in future studies.
Keywords: intestinal failure; parenteral nutrition; short bowel syndrome; survey; teduglutide.
Conflict of interest statement
F.H. received a grant and lecture fee from Takeda outside of the submitted work. S.M. and P.B. are employees of Takeda Pharmaceuticals International AG and hold Takeda stock or stock options. L.H. is an employee of Adelphi Real World, which received payment from Takeda Pharmaceuticals International AG as part of this research. U.-F.P. received consultation/lecture/advisory board honoraria and grant/research support from Takeda/Shire Pharmaceuticals/NPS Pharmaceuticals, received research support from Hanmi Pharma and Veciv Bio Pharma, and received consultation/advisory board honoraria and research support from Zealand Pharma.
Figures
Similar articles
-
Independence From Parenteral Nutrition and Intravenous Fluid Support During Treatment With Teduglutide Among Patients With Intestinal Failure Associated With Short Bowel Syndrome.JPEN J Parenter Enteral Nutr. 2017 Aug;41(6):946-951. doi: 10.1177/0148607116680791. Epub 2016 Nov 23. JPEN J Parenter Enteral Nutr. 2017. PMID: 27875291 Free PMC article. Clinical Trial.
-
Long-Term Therapy With Teduglutide in Parenteral Support-Dependent Patients With Short Bowel Syndrome: A Case Series.JPEN J Parenter Enteral Nutr. 2018 May;42(4):821-825. doi: 10.1002/jpen.1149. Epub 2018 Mar 30. JPEN J Parenter Enteral Nutr. 2018. PMID: 29603279
-
Factors Associated With Response to Teduglutide in Patients With Short-Bowel Syndrome and Intestinal Failure.Gastroenterology. 2018 Mar;154(4):874-885. doi: 10.1053/j.gastro.2017.11.023. Epub 2017 Nov 22. Gastroenterology. 2018. PMID: 29174926 Clinical Trial.
-
Teduglutide for the treatment of short bowel syndrome - a safety evaluation.Expert Opin Drug Saf. 2018 Jul;17(7):733-739. doi: 10.1080/14740338.2018.1483332. Epub 2018 Jun 8. Expert Opin Drug Saf. 2018. PMID: 29848084 Review.
-
Teduglutide: a guide to its use in short bowel syndrome.Clin Drug Investig. 2015 May;35(5):335-40. doi: 10.1007/s40261-015-0286-6. Clin Drug Investig. 2015. PMID: 25859983 Review.
References
-
- Bioletto F., D‘Eusebio C., Merlo F.D., Aimasso U., Ossola M., Pellegrini M., Ponzo V., Chiarotto A., De Francesco A., Ghigo E., et al. Efficacy of teduglutide for parenteral support reduction in patients with short bowel syndrome: A systematic review and meta-analysis. Nutrients. 2022;14:796. doi: 10.3390/nu14040796. - DOI - PMC - PubMed
-
- Büttner J., Blüthner E., Greif S., Kühl A., Elezkurtaj S., Ulrich J., Maasberg S., Jochum C., Tacke F., Pape U.F. Predictive potential of biomarkers of intestinal barrier function for therapeutic management with teduglutide in patients with short bowel syndrome. Nutrients. 2023;15:4220. doi: 10.3390/nu15194220. - DOI - PMC - PubMed
-
- Pevny S., Maasberg S., Rieger A., Karber M., Blüthner E., Knappe-Drzikova B., Thurmann D., Büttner J., Weylandt K.H., Wiedenmann B., et al. Experience with teduglutide treatment for short bowel syndrome in clinical practice. Clin. Nutr. 2019;38:1745–1755. doi: 10.1016/j.clnu.2018.07.030. - DOI - PubMed
-
- Berghöfer P., Fragkos K.C., Baxter J.P., Forbes A., Joly F., Heinze H., Loth S., Pertkiewicz M., Messing B., Jeppesen P.B. Development and validation of the disease-specific Short Bowel Syndrome-Quality of Life (SBS-QoL™) scale. Clin. Nutr. 2013;32:789–796. doi: 10.1016/j.clnu.2012.12.001. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

