Activation of α2B/2C adrenergic receptor ameliorates ocular surface inflammation through enhancing regulatory T cell function
- PMID: 39522611
- PMCID: PMC11869812
- DOI: 10.1016/j.mucimm.2024.11.002
Activation of α2B/2C adrenergic receptor ameliorates ocular surface inflammation through enhancing regulatory T cell function
Abstract
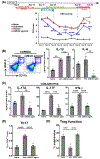
There is an unmet need for effectively treating dry eye disease (DED), a T cell-mediated chronic, inflammatory ocular surface disorder. Given the potential of nonneuronal adrenergic system in modulating T cell response, we herein investigated the therapeutic efficacy and the underlying mechanisms of a specific alpha 2 adrenergic receptor agonist (AGN-762, selective for α2B/2C receptor subtypes) in a mouse model of DED. Experimental DED was treated with the AGN-762 by oral gavage, either at disease induction or after disease establishment, and showed sustained amelioration, along with reduced expression of DED-pathogenic cytokines in ocular surface tissues, decreased corneal MHC-II+CD11b+ cells and lymphoid Th17 cells, and higher function of regulatory T cells (Treg). In vitro culture of DED-derived effector T helper cells (Teff) with AGN-762 failed to suppress Th17 response, while culture of DED-Treg with AGN-762 led to enhanced suppressive function of Treg and their IL-10 production. Adoptive transfer of AGN-762-pretreated DED-Treg in syngeneic B6.Rag1-/- mice effectively suppressed DED Teff-mediated disease and Th17 response, and the effect was abolished by the neutralization of IL-10. In conclusion, our findings demonstrate that α2B/2C adrenergic receptor agonism effectively ameliorates persistent corneal epitheliopathy in DED by enhancing IL-10 production from Treg and thus restoring their immunoregulatory function.
Copyright © 2024 The Author(s). Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of competing interest The authors declare the following financial interests/personal relationships which may be considered as potential competing interests: This work was supported by a sponsored research award from AbbVie Inc. to Reza Dana and Yihe Chen. Veena Viswanath and Daniel Gil are former employees of Allergan Inc./AbbVie Inc.. Katherine Held is a current employee of AbbVie Inc.
Figures






References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

