Spermine oxidase promotes Helicobacter pylori-mediated gastric carcinogenesis through acrolein production
- PMID: 39523394
- PMCID: PMC11779639
- DOI: 10.1038/s41388-024-03218-7
Spermine oxidase promotes Helicobacter pylori-mediated gastric carcinogenesis through acrolein production
Abstract
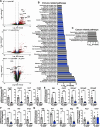
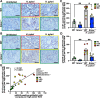
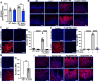
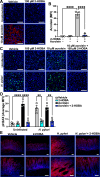
Helicobacter pylori is the primary cause of gastric cancer, and there is a need to discover new molecular targets for therapeutic intervention in H. pylori disease progression. We have previously shown that spermine oxidase (SMOX), the enzyme that catabolizes the back-conversion of the polyamine spermine to spermidine, is upregulated during infection and is associated with increased cancer risk in humans. We sought to determine the direct role of SMOX in gastric carcinogenesis during H. pylori infection. In this study, we demonstrate that transgenic FVB/N insulin-gastrin (INS-GAS) mice that develop gastric carcinoma with H. pylori infection were protected from cancer development with Smox deletion. RNA sequencing revealed that genes associated with the immune system and cancer were downregulated in the infected Smox-/- mice. Furthermore, there was a decrease in cell proliferation and DNA damage in infected Smox-/- animals. There was significant generation of adducts of the highly reactive electrophile acrolein, a byproduct of SMOX activity, in gastric tissues from H. pylori-infected humans and wild-type, but not Smox-/- mice. Genetic deletion of Smox in murine organoids or chemical inhibition of SMOX in human gastric epithelial cells significantly reduced generation of acrolein induced by H. pylori. Additionally, acrolein-induced DNA damage in gastric epithelial cells was ablated with the electrophile scavenger 2-hydroxybenzylamine (2-HOBA). Gastric acrolein adduct levels were attenuated in infected INS-GAS mice treated with 2-HOBA, which exhibit reduced gastric carcinoma. These findings implicate SMOX and acrolein in H. pylori-induced carcinogenesis, thus indicating their potential as therapeutic targets.
© 2024. This is a U.S. Government work and not under copyright protection in the US; foreign copyright protection may apply.
Conflict of interest statement
Competing interests: APG and KTW are named inventors on a US patent, Compounds and Methods for Scavenging Dicarbonyl Electrophiles (Patent # 11696903), for use of electrophile scavengers, which is assigned to Vanderbilt University and The United States Government as represented by the Department of Veterans Affairs. In addition, APG and KTW receive royalty payments from a licensing agreement between Vanderbilt University and MTI Biotech for the future use of electrophile scavengers. All other authors have declared that no conflict of interest exists.
Figures

References
-
- Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229–63. - PubMed
-
- Plummer M, Franceschi S, Vignat J, Forman D, de Martel C. Global burden of gastric cancer attributable to Helicobacter pylori. Int J Cancer. 2015;136:487–90. - PubMed
-
- Hooi JKY, Lai WY, Ng WK, Suen MMY, Underwood FE, Tanyingoh D, et al. Global prevalence of Helicobacter pylori infection: systematic review and meta-analysis. Gastroenterology. 2017;153:420–9. - PubMed
-
- Correa P. Human gastric carcinogenesis: a multistep and multifactorial process-First American Cancer Society Award Lecture on Cancer Epidemiology and Prevention. Cancer Res. 1992;52:6735–40. - PubMed
-
- Correa P, Shiao YH. Phenotypic and genotypic events in gastric carcinogenesis. Cancer Res. 1992;54:1941s–1943s. - PubMed
MeSH terms
Substances
Grants and funding
- R01 DK128200/DK/NIDDK NIH HHS/United States
- I01CX002171/U.S. Department of Veterans Affairs (Department of Veterans Affairs)
- P30 DK058404/DK/NIDDK NIH HHS/United States
- T32 AI138932/AI/NIAID NIH HHS/United States
- T32CA009592/U.S. Department of Health & Human Services | NIH | National Cancer Institute (NCI)
- W81XWH-18-1-0301/U.S. Department of Defense (United States Department of Defense)
- P01CA116087/U.S. Department of Health & Human Services | NIH | National Cancer Institute (NCI)
- I01 CX002171/CX/CSRD VA/United States
- F31 CA278330/CA/NCI NIH HHS/United States
- R01DK128200/U.S. Department of Health & Human Services | NIH | National Institute of Diabetes and Digestive and Kidney Diseases (National Institute of Diabetes & Digestive & Kidney Diseases)
- IK2BX004748/U.S. Department of Veterans Affairs (Department of Veterans Affairs)
- IK2 BX004648/BX/BLRD VA/United States
- F31CA278330/U.S. Department of Health & Human Services | NIH | National Cancer Institute (NCI)
- I01CX002473/U.S. Department of Veterans Affairs (Department of Veterans Affairs)
- W81XWH-21-1-0617/U.S. Department of Defense (United States Department of Defense)
- R41CA257262/U.S. Department of Health & Human Services | NIH | National Cancer Institute (NCI)
- I01BX001453/U.S. Department of Veterans Affairs (Department of Veterans Affairs)
- P01 CA028842/CA/NCI NIH HHS/United States
- I01 CX002473/CX/CSRD VA/United States
- I01 CX002662/CX/CSRD VA/United States
- P01 CA116087/CA/NCI NIH HHS/United States
- P01CA028842/U.S. Department of Health & Human Services | NIH | National Cancer Institute (NCI)
- I01BX004366/U.S. Department of Veterans Affairs (Department of Veterans Affairs)
- R21AI142042/U.S. Department of Health & Human Services | NIH | National Institute of Allergy and Infectious Diseases (NIAID)
- I01 BX001453/BX/BLRD VA/United States
- T32AI138932/U.S. Department of Health & Human Services | NIH | National Institute of Allergy and Infectious Diseases (NIAID)
- I01 BX004366/BX/BLRD VA/United States
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

