Macrophages producing chondroitin sulfate proteoglycan-4 induce neuro-cardiac junction impairment in Duchenne muscular dystrophy
- PMID: 39523812
- PMCID: PMC11638662
- DOI: 10.1002/path.6362
Macrophages producing chondroitin sulfate proteoglycan-4 induce neuro-cardiac junction impairment in Duchenne muscular dystrophy
Abstract
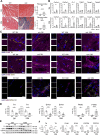
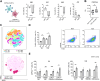
Duchenne muscular dystrophy (DMD) is caused by the absence of the full form of the dystrophin protein, which is essential for maintaining the structural integrity of muscle cells, including those in the heart and respiratory system. Despite progress in understanding the molecular mechanisms associated with DMD, myocardial insufficiency persists as the primary cause of mortality, and existing therapeutic strategies remain limited. This study investigates the hypothesis that a dysregulation of the biological communication between infiltrating macrophages (MPs) and neurocardiac junctions exists in dystrophic cardiac tissue. In a mouse model of DMD (mdx), this phenomenon is influenced by the over-release of chondroitin sulfate proteoglycan-4 (CSPG4), a key inhibitor of nerve sprouting and a modulator of the neural function, by MPs infiltrating the cardiac tissue and associated with dilated cardiomyopathy, a hallmark of DMD. Givinostat, the histone deacetylase inhibitor under current development as a clinical treatment for DMD, is effective at both restoring a physiological microenvironment at the neuro-cardiac junction and cardiac function in mdx mice in addition to a reduction in cardiac fibrosis, MP-mediated inflammation, and tissue CSPG4 content. This study provides novel insight into the pathophysiology of DMD in the heart, identifying potential new biological targets. © 2024 The Author(s). The Journal of Pathology published by John Wiley & Sons Ltd on behalf of The Pathological Society of Great Britain and Ireland.
Keywords: DMD heart failure; cardiac fibrosis; cardiac innervation; dilated cardiomyopathy; extracellular matrix; inflammation; macrophages; proteoglycans.
© 2024 The Author(s). The Journal of Pathology published by John Wiley & Sons Ltd on behalf of The Pathological Society of Great Britain and Ireland.
Figures




References
-
- Lombardi L, Stefano MED, Paggi P. Components of the NGF signaling complex are altered in mdx mouse superior cervical ganglion and its target organs. Neurobiol Dis 2008; 32: 402–411. - PubMed
-
- Shannon TR. Ryanodine receptor Ca2+ sensitivity and excitation‐contraction coupling in muscular dystrophy and heart failure: similar and yet different. Am J Physiol Heart Circ Physiol 2009; 297: H1965–H1966. - PubMed
-
- Canonico F, Chirivi M, Maiullari F, et al. Focus on the road to modelling cardiomyopathy in muscular dystrophy. Cardiovasc Res 2022; 118: 1872–1884. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

