Palmitoylcarnitine impairs immunity in decompensated cirrhosis
- PMID: 39524205
- PMCID: PMC11544064
- DOI: 10.1016/j.jhepr.2024.101187
Palmitoylcarnitine impairs immunity in decompensated cirrhosis
Abstract
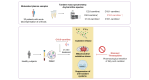
Background & aims: In patients with cirrhosis, acute decompensation (AD) correlates with a hyperinflammatory state driven by mitochondrial dysfunction, which is a significant factor in the progression toward acute-on-chronic liver failure (ACLF). Elevated circulating levels of acylcarnitine, indicative of mitochondrial dysfunction, are predictors of mortality in ACLF patients. Our hypothesis posits that acylcarnitines not only act as biomarkers, but also actively exert detrimental effects on circulating immune cells.
Methods: Plasma acylcarnitine levels were measured in 20 patients with AD cirrhosis and 10 healthy individuals. The effects of selected medium- and long-chain acylcarnitines on mitochondrial function were investigated in peripheral leucocytes from healthy donors by determining mitochondrial membrane potential (Δψm) and mitochondrial respiration using the JC-1 dye and Agilent Seahorse XF technology. Changes regarding mitochondrial ultrastructure and redox systems were assessed by transmission electron microscopy and gene and protein expression analysis.
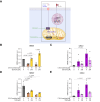
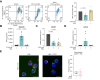
Results: Plasma levels of several acylcarnitine species were significantly elevated in patients with AD cirrhosis compared with healthy individuals, alongside increased levels of inflammatory mediators (cytokines and chemokines). Notably, the long-chain acylcarnitine palmitoylcarnitine (C16:0-carnitine, 1.51-fold higher, p = 0.0059) impaired Δψm and reduced the spare respiratory capacity of peripheral mononuclear leucocytes. Additionally, C16:0-carnitine induced mitochondrial oxidative stress, suppressed the expression of the antioxidant gene HMOX1, and increased CXCL8 expression and IL-8 release. Etomoxir, which blocks acylcarnitine entry into the mitochondria, reversed the suppression of HMOX1. Similarly, trimetazidine, a fatty acid beta-oxidation inhibitor, prevented C16:0-carnitine-induced CXCL8 expression. Importantly, oxidative stress and Δψm impairment caused by C16:0-carnitine were less severe in the presence of albumin, a standard therapy for AD cirrhosis.
Conclusions: Our findings suggest that long-chain acylcarnitines induce mitochondrial injury in immune cells, thereby contributing to the development of immune dysfunction associated with cirrhosis.
Impact and implications: Patients with acute decompensation of cirrhosis and acute-on-chronic liver failure (ACLF) display a systemic hyperinflammatory state and leukocyte mitochondrial dysfunction. We discovered that apart from being increased in the circulation of these patients, the long-chain palmitoylcarnitine is able to elicit cytokine secretion paired with mitochondrial dysfunction in leukocytes from healthy donors. In particular, we show that inhibiting the metabolism of palmitoylcarnitine could reverse these detrimental effects. Our findings underline the importance of immunometabolism as a treatment target in patients with acute decompensation of cirrhosis and ACLF.
Keywords: Acute decompensation of cirrhosis; Acylcarnitines; Immune cells; Mitochondrial dysfunction.
© 2024 The Authors.
Figures




References
-
- McGarry J.D., Brown N.F. The mitochondrial carnitine palmitoyltransferase system. From concept to molecular analysis. Eur J Biochem. 1997;244:1–14. - PubMed
-
- Reuter S.E., Evans A.M. Carnitine and acylcarnitines: pharmacokinetic, pharmacological and clinical aspects. Clin Pharmacokinet. 2012;51:553–572. - PubMed

