Increased muscle coactivation is linked with fast feedback control when reaching in unpredictable visual environments
- PMID: 39524350
- PMCID: PMC11550142
- DOI: 10.1016/j.isci.2024.111174
Increased muscle coactivation is linked with fast feedback control when reaching in unpredictable visual environments
Abstract
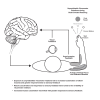
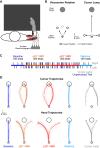
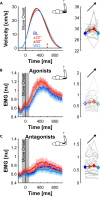
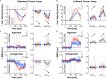
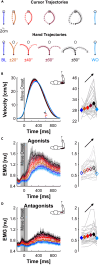
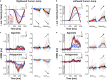
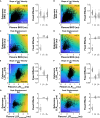
Humans encounter unpredictable disturbances in daily activities and sports. When encountering unpredictable physical disturbances, healthy participants increase the peak velocity of their reaching movements, muscle coactivation, and responses to sensory feedback. Emerging evidence suggests that muscle coactivation may facilitate responses to sensory feedback and may not solely increase stiffness to resist displacements. We tested this idea by examining how healthy participants alter the control of reaching movements and responses to sensory feedback when encountering variable visuomotor rotations. The rotations changed amplitude and direction between movements, creating unpredictable errors that required fast online corrections. Participants increased the peak velocity of their movements, muscle coactivation, and responses to visual and proprioceptive feedback with the variability of the visuomotor rotations. The findings highlight an increase in neural responsiveness to sensory feedback and suggest that muscle coactivation may prime the nervous system for fast responses to sensory feedback that accommodate properties of unpredictable visual environments.
Keywords: cognitive neuroscience; neuroscience; sensory neuroscience.
© 2024 The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures

Similar articles
-
The nervous system tunes sensorimotor gains when reaching in variable mechanical environments.iScience. 2023 Apr 27;26(6):106756. doi: 10.1016/j.isci.2023.106756. eCollection 2023 Jun 16. iScience. 2023. PMID: 37213228 Free PMC article.
-
Robust Control in Human Reaching Movements: A Model-Free Strategy to Compensate for Unpredictable Disturbances.J Neurosci. 2019 Oct 9;39(41):8135-8148. doi: 10.1523/JNEUROSCI.0770-19.2019. Epub 2019 Sep 5. J Neurosci. 2019. PMID: 31488611 Free PMC article.
-
Online corrective responses following target jump in altered gravitoinertial force field point to nested feedforward and feedback control.J Neurophysiol. 2021 Jan 1;125(1):154-165. doi: 10.1152/jn.00268.2020. Epub 2020 Nov 11. J Neurophysiol. 2021. PMID: 33174494
-
A perspective on multisensory integration and rapid perturbation responses.Vision Res. 2015 May;110(Pt B):215-22. doi: 10.1016/j.visres.2014.06.011. Epub 2014 Jul 9. Vision Res. 2015. PMID: 25014401 Review.
-
The control and perception of antagonist muscle action.Exp Brain Res. 2023 Jan;241(1):1-12. doi: 10.1007/s00221-022-06498-1. Epub 2022 Oct 30. Exp Brain Res. 2023. PMID: 36309879 Review.
Cited by
-
Success-efficient/failure-safe strategy for hierarchical reinforcement motor learning.PLoS Comput Biol. 2025 May 9;21(5):e1013089. doi: 10.1371/journal.pcbi.1013089. eCollection 2025 May. PLoS Comput Biol. 2025. PMID: 40344154 Free PMC article.
References
-
- Franklin S., Franklin D.W. Feedback Gains modulate with Motor Memory Uncertainty. Neuron. Behav. Data Anal. Theory. 2021;5:1–28. doi: 10.51628/001c.22336. - DOI
-
- Franklin D.W., Burdet E., Osu R., Kawato M., Milner T.E. Functional significance of stiffness in adaptation of multijoint arm movements to stable and unstable dynamics. Exp. Brain Res. 2003;151:145–157. - PubMed
LinkOut - more resources
Full Text Sources

