Local and systemic humoral immune responses to Histophilus somni recombinant antigens administered intranasally and subcutaneously to dairy calves
- PMID: 39528575
- PMCID: PMC11555116
- DOI: 10.1038/s41598-024-78605-x
Local and systemic humoral immune responses to Histophilus somni recombinant antigens administered intranasally and subcutaneously to dairy calves
Abstract
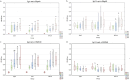
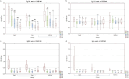
Bovine respiratory disease (BRD) causes significant economic losses in dairy calves. Induction of an early immune response via parenteral vaccination is complicated by the interference of colostral immunity. In this study, we investigated early immunization against selected conserved bacterial antigens. Calves were vaccinated twice intranasally and then subcutaneously with Histophilus somni recombinant proteins (rOMP40 or rHsp60) mixed with one of two adjuvants: CpG ODN2007 or MPLA. The control group (Con) was treated with PBS. The first immunization was done between 24 and 48 h of life and then twice in two weeks intervals. Blood, nasal, and saliva secretion samples were collected directly before vaccination (S1-S3) and then on 42-44 (S4) and 59-61 (S5) day of life. Antibodies (IgG1/IgG2/IgM/IgA in serum; IgG1/IgA in secretions) against both vaccine antigens were quantified in all samples. Intranasal and subcutaneous vaccinations using the described formulas did not increase antibody reactivity against the tested proteins. The reactivity of serum IgG1, IgM, and IgA anti-rOMP40 antibodies was significantly higher in S1 in all groups than that in the other samplings (p˂0.01). Significant differences in the reactivity of serum anti-rOMP40 antibodies between groups were identified in S1 (IgA reactivity was higher in the CpG vs. MPLA group; p < 0.05), S4 (IgM reactivity was higher in Con vs. CpG group; p < 0.05), and S5 (IgG1 reactivity was higher in MPLA vs. Con group; p < 0.05). The lack of consistent changes in antibodies after immunization (S4 and S5) hinders the drawing of conclusions regarding the effect of immunization on antibody reactivity. In the future, establishing a proper immunization window and adjuvants for nasal vaccines against bacterial pathogens causing BRD in calves remains to be determined.
Keywords: CpG ODN2007; MPLA; rHsp60 H. somni; rOMP40 H. somni.
© 2024. The Author(s).
Conflict of interest statement
Figures

Similar articles
-
IbpA DR2 subunit immunization protects calves against Histophilus somni pneumonia.Vaccine. 2011 Jun 24;29(29-30):4805-12. doi: 10.1016/j.vaccine.2011.04.075. Epub 2011 May 8. Vaccine. 2011. PMID: 21557979
-
Subcutaneous application of hyperimmune serum against Histophilus somni recombinant proteins affects serum antibody reactivity in beef calves.BMC Vet Res. 2024 Feb 10;20(1):51. doi: 10.1186/s12917-024-03895-2. BMC Vet Res. 2024. PMID: 38341558 Free PMC article.
-
Antibody responses of calves to Histophilus somni recombinant IbpA subunits.Comp Immunol Microbiol Infect Dis. 2012 Sep;35(5):453-9. doi: 10.1016/j.cimid.2012.04.002. Epub 2012 May 2. Comp Immunol Microbiol Infect Dis. 2012. PMID: 22554920
-
Precolostral antibody reactivity against Histophilus somni conserved proteins and Escherichia coli whole cells in stillborn and live calves.Immunobiology. 2025 Mar;230(2):152874. doi: 10.1016/j.imbio.2025.152874. Epub 2025 Jan 26. Immunobiology. 2025. PMID: 39891975
-
Host Immune Response to Histophilus somni.Curr Top Microbiol Immunol. 2016;396:109-29. doi: 10.1007/82_2015_5012. Curr Top Microbiol Immunol. 2016. PMID: 26728062 Review.
Cited by
-
Hyperimmune serum containing Histophilus somni rHsp60, rOMP40 and Actinobacillus pleuropneumoniae LPS antibodies as supplementary treatment for calf respiratory diseases.Sci Rep. 2025 May 30;15(1):19088. doi: 10.1038/s41598-025-03624-1. Sci Rep. 2025. PMID: 40447709 Free PMC article.
References
-
- Osman, R., Malmuthuge, N. & Griebel, P. G. C. P. Development and function of the Mucosal Immune System in the Upper respiratory tract of neonatal calves. Annu. Rev. Anim. Biosci.6, 141–155 (2017). - PubMed
-
- Jain, R., Waldvogel-Thurlow, S., Darveau, R. & Douglas, R. Differences in the paranasal sinuses between germ-free and pathogen-free mice. Int. Forum Allergy Rhinol. 6, 631–637 (2016). - PubMed
-
- Wu, H. Y., Nguyen, H. H. & Russell, M. W. Nasal lymphoid tissue (NALT) as a mucosal immune inductive site. Scand. J. Immunol.46, 506–513 (1997). - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

