Drug prioritization identifies panobinostat as a tailored treatment element for patients with metastatic hepatoblastoma
- PMID: 39529166
- PMCID: PMC11556140
- DOI: 10.1186/s13046-024-03221-6
Drug prioritization identifies panobinostat as a tailored treatment element for patients with metastatic hepatoblastoma
Abstract
Background: Patients with metastatic hepatoblastoma are treated with severely toxic first-line chemotherapies in combination with surgery. Yet, inadequate response of lung metastases to neo-adjuvant chemotherapy still compromises patient outcomes making new treatment strategies, tailored to more efficient lung clearance, mandatory.
Methods: We harnessed a comprehensive patient-derived xenograft platform and a variety of in vitro and in vivo assays to establish the preclinical and biological rationale for a new drug for patients with metastatic hepatoblastoma.
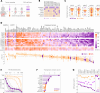
Results: The testing of a library of established drugs on patient-derived xenografts identified histone deacetylase inhibitors, most notably panobinostat, to be highly efficacious on hepatoblastoma cells, as compared to non-cancerous cells. Molecularly, the anti-tumor effect of panobinostat is mediated by posttranslational obstruction of the MYC oncoprotein as a result of dual specificity phosphatase 1 upregulation, thereby leading to growth inhibition and programmed cell death. Of clinical importance, upregulation of the MYC target gene nucleophosmin 1 is indicative of response to panobinostat and associated with metastatic disease in patients with hepatoblastoma. The combination of panobinostat with the current SIOPEL 4 induction protocol, consisting of cisplatin and doxorubicin, revealed high synergies already at low nanomolar levels. The simulation of a clinical trial, with this combination therapy, in patient-derived xenograft models, and ultimately heterotypic lung metastasis mimics clearly underscored the potency of this approach.
Conclusion: Integrated studies define MYC inhibition by panobinostat as a novel treatment element to be introduced into the therapeutic strategy for patients with metastatic hepatoblastoma.
Keywords: Hepatoblastoma; MYC; Metastasis; Panobinostat; Therapy.
© 2024. The Author(s).
Conflict of interest statement
Figures





References
-
- Weinberg AG, Finegold MJ. Primary hepatic tumors of childhood. Hum Pathol. 1983;14(6):512–37. - PubMed
-
- Perilongo G, Maibach R, Shafford E, Brugieres L, Brock P, Morland B, et al. Cisplatin versus cisplatin plus doxorubicin for standard-risk hepatoblastoma. N Engl J Med. 2009;361(17):1662–70. - PubMed
-
- Zsiros J, Maibach R, Shafford E, Brugieres L, Brock P, Czauderna P, et al. Successful treatment of childhood high-risk hepatoblastoma with dose-intensive multiagent chemotherapy and surgery: final results of the SIOPEL-3HR study. J Clin Oncol. 2010;28(15):2584–90. - PubMed
-
- van Kalsbeek RJ, Hudson MM, Mulder RL, Ehrhardt M, Green DM, Mulrooney DA, et al. A joint international consensus statement for measuring quality of survival for patients with childhood cancer. Nat Med. 2023;29(6):1340–8. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

