Liver transplantation for critically ill patients with acute on chronic liver failure: a prospective national programme of waitlist prioritisation
- PMID: 39529808
- PMCID: PMC11551510
- DOI: 10.1016/j.lanepe.2024.101067
Liver transplantation for critically ill patients with acute on chronic liver failure: a prospective national programme of waitlist prioritisation
Abstract
Background: Acute on Chronic Liver Failure (ACLF) complicates chronic liver disease (CLD) combining rapidly progressive hepatic with extra-hepatic multiple organ failure and high short-term mortality. Effective therapeutic options are very limited, and liver transplantation (LT) seldom utilised through concerns of high recipient mortality and resource use. Retrospective reports suggest recent outcomes may have improved, but use of LT for ACLF has not been prospectively assessed.
Methods: A prospective programme of prioritised liver graft allocation for selected recipients with ACLF through registration on a new national tier, initiated in May 2021 in all 7 United Kingdom LT centres. Candidates were selected by centre multidisciplinary teams, with inclusion criteria mandating cirrhotic CLD with ACLF requiring critical care (CC) organ support and expected 1-month mortality >50%. Exclusion criteria included age ≥60 years, previous LT, comorbidity or substance misuse profile precluding elective LT. A pilot 50 registrations were planned, with pre-specified futility criteria of a 1-year post-LT survival of 60%.
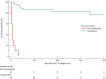
Findings: Fifty-two patients were registered on the ACLF tier, median (IQR) age 46 (39-52) years, ACLF grade 3 (3-3) and Model for End-stage Liver Disease (MELD) 39 (35-40). At registration 32 (62%) required mechanical ventilation, 44 (85%) vasopressors and 46 (89%) renal replacement. Forty-two (81%) underwent LT 2 (2-5) days after registration: 10 (19%) did not. All non-transplanted died at median 7 (4-13) days after registration (p < 0.0001 vs. LT). Post-LT follow-up was 212 (119-530) days and patient survival 81% (95% CI 66-91): 28-, 90-day and 1-year survival after registration 93%, 86% and 77%. Median length of CC and hospital stay in LT recipients was 16 (8-28) and 35 (23-54) days respectively.
Interpretation: We report the first prospective national series of prioritised liver transplantation for critically ill patients with ACLF. For selected recipients LT is a practical and highly effective treatment option where no other similarly effective interventions exist.
Funding: There was no funding for the study.
Keywords: Acute on chronic liver failure; Critical care; Liver transplantation.
© 2024 The Authors.
Conflict of interest statement
WB: Consulting Fees: Sana Biotech/Flagship Pioneering, RT: none, IAR: Consulting Fees: Roche, Novo Nordisk, Boehringer Ingelheim, Honoraria: Norgine, Research Committee Chair, British Association for the Study of the Liver, AC: none, MJA: none, MEDA: Payment or honoraria: GSK: made to CUHFT as part of research collaboration, GW: Support for attending meetings from NHS Blood and Transplant, TP: none, JM: none, LB: none, SM: none, DC: none, BJH: none, RW: none, RJ: Royalties or Licenses: Co-Founder (with stock options): Yaqrit Ltd, Cyberliver Limited, Hepyx, Limited, Payment or honoraria: Grifols, Committee evaluating grants, Patents planned, issued or pending: Patent Families: DIALIVE, CARBALIVE, TLR4 antagonists, Targeting Pyroptosis; Targeting Necroptosis; Ornithine Phenylacetate, KJS: none, JI: none, DT: none.
Figures
References
LinkOut - more resources
Full Text Sources