C3 glomerulopathy: a kidney disease mediated by alternative pathway deregulation
- PMID: 39534179
- PMCID: PMC11554616
- DOI: 10.3389/fneph.2024.1460146
C3 glomerulopathy: a kidney disease mediated by alternative pathway deregulation
Abstract
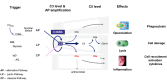
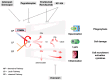
C3 glomerulopathy (C3G) is an ultra-rare complement-mediated kidney disease caused by to the deregulation of the alternative pathway (AP) of proximal complement. Consequently, all effector loops of the complement are active and can lead to pathologies, such as C3a- and C5a-mediated inflammation, C3b opsonization, surface C3b-mediated AP C3 convertase assembly, C3 cleavage product deposition in the glomerulus, and lytic C5b-9/MAC cell damage. The most common pathologic mechanisms are defective chronic alternative pathway deregulation, mostly occurring in the plasma, often causing C3 consumption, and chronic complement-mediated glomerular damage. C3G develops over several years, and loss of renal function occurs in more than 50% of patients. C3G is triggered by both genetic and autoimmune alterations. Genetic causes include mutations in individual complement genes and chromosomal variations in the form of deletions and duplications affecting genes encoding complement modulators. Many genetic aberrations result in increased AP C3 convertase activity, either due to decreased activity of regulators, increased activity of modulators, or gain-of-function mutations in genes encoding components of the convertase. Autoimmune forms of C3G do also exist. Autoantibodies target individual complement components and regulators or bind to neoepitopes exposed in the central alternative pathway C3 convertase, thereby increasing enzyme activity. Overactive AP C3 convertase is common in C3G patients. Given that C3G is a complement disease mediated by defective alternative pathway action, complement blockade is an emerging concept for therapy. Here, we summarize both the causes of C3G and the rationale for complement inhibition and list the inhibitors that are being used in the most advanced clinical trials for C3G. With several inhibitors in phase II and III trials, it is expected that effectice treatment for C3G will become availabe in the near future.
Keywords: C3 glomerulopathy; alternative pathway (AP); complement; complement inhibitors; therapy.
Copyright © 2024 Heidenreich, Goel, Priyamvada, Kulkarni, Chakurkar, Khullar, Singh, Bale and Zipfel.
Conflict of interest statement
KH is employed by Eleva GmbH. PFZ received funding from the DFG priority program SFB 1192, Immune-mediated glomerular diseases project B6, Kidneeds and consulting fees from Alnylam, Bayer, Novartis, Samsung Bioepis, Alexion/AstraZeneca Rare Disease, CSL Vifor, and Eleva GmbH. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Figures


References
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

