Assessment of the Efficacy of Deep Brain Stimulation (DBS) in Managing Drug-Resistant Epilepsy (DRE): A Systematic Review and Meta-Analysis of Randomized Controlled Trials (RCTs)
- PMID: 39534832
- PMCID: PMC11555489
- DOI: 10.7759/cureus.71348
Assessment of the Efficacy of Deep Brain Stimulation (DBS) in Managing Drug-Resistant Epilepsy (DRE): A Systematic Review and Meta-Analysis of Randomized Controlled Trials (RCTs)
Abstract
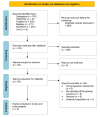
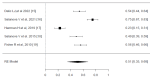
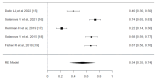
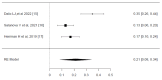
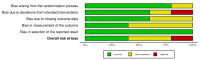
Epilepsy is a chronic neurological disorder affecting millions of people around the world. Even though the majority of patients gain seizure control with antiseizure medications (ASMs), many subjects may have drug-resistant epilepsy (DRE). Deep brain stimulation (DBS) is a promising alternative, showing effectiveness in reducing seizures for some patients. This systematic review and meta-analysis aim to evaluate DBS's efficacy in DRE. A comprehensive search in PubMed, CENTRAL, Medline, Ovid, and Scopus was performed up to August 31, 2024, using the terms 'Drug-resistant Epilepsy' AND 'Deep Brain Stimulation'. Two independent researchers screened titles, abstracts, and full texts. The data extracted included study details, sample size, age at surgery, seizure duration, follow-up duration, seizure reduction (SR), responder rate (RR), and adverse events. Quality assessment was conducted using the Risk of Bias 2 (ROB2) tool (https://methods.cochrane.org/bias/resources/rob-2-revised-cochrane-risk-bias-tool-randomized-trials), and data analysis was performed using Jamovi software (https://www.jamovi.org). The search yielded 707 studies that were initially screened. Out of them, 29 articles were retrieved for full-text screening, and 5 randomized controlled trials (RCTs) were included in the review and meta-analysis. The meta-analysis showed that the pooled effect size for SR was 0.51 (95% CI, 0.35-0.68; P < 0.001), and the pooled effect size for RR was 0.54 (95% CI, 0.35-0.74; P < 0.001), demonstrating significant improvements in seizure control. The pooled effect size for adverse events was 0.21 (95% CI, 0.08-0.34; P = 0.001). The risk-of-bias assessment revealed a low risk of randomization for most studies. However, concerns were noted in areas such as deviations from the intended intervention and missing outcome data. In conclusion, DBS is a viable intervention for DRE, with significant reductions in seizure frequency and a favorable safety profile. However, the variability in efficacy and RRs across studies underscores the need for continued research to refine patient selection criteria, optimize stimulation parameters, and explore the differential effects of targeting various thalamic nuclei.
Keywords: deep brain stimulation; drug-resistant epilepsy; meta-analysis; responder rate; seizure reduction.
Copyright © 2024, Raslan et al.
Conflict of interest statement
Conflicts of interest: In compliance with the ICMJE uniform disclosure form, all authors declare the following: Payment/services info: All authors have declared that no financial support was received from any organization for the submitted work. Financial relationships: All authors have declared that they have no financial relationships at present or within the previous three years with any organizations that might have an interest in the submitted work. Other relationships: All authors have declared that there are no other relationships or activities that could appear to have influenced the submitted work.
Figures

Similar articles
-
Deep brain stimulation targets in drug-resistant epilepsy: Systematic review and meta-analysis of effectiveness and predictors of response.Seizure. 2024 Nov;122:144-152. doi: 10.1016/j.seizure.2024.09.017. Epub 2024 Sep 29. Seizure. 2024. PMID: 39423756
-
A systematic review of deep brain stimulation for the treatment of drug-resistant epilepsy in childhood.J Neurosurg Pediatr. 2019 Mar 1;23(3):274-284. doi: 10.3171/2018.9.PEDS18417. Epub 2018 Nov 30. J Neurosurg Pediatr. 2019. PMID: 30544364
-
Deep brain stimulation for epilepsy: A systematic review and meta-analysis of randomized and non-randomized studies of thalamic targeting.Epilepsy Res. 2025 Jun 7;216:107607. doi: 10.1016/j.eplepsyres.2025.107607. Online ahead of print. Epilepsy Res. 2025. PMID: 40516441 Review.
-
Treatments for seizures in catamenial (menstrual-related) epilepsy.Cochrane Database Syst Rev. 2019 Oct 14;10(10):CD013225. doi: 10.1002/14651858.CD013225.pub2. Cochrane Database Syst Rev. 2019. Update in: Cochrane Database Syst Rev. 2021 Sep 16;9:CD013225. doi: 10.1002/14651858.CD013225.pub3. PMID: 31608992 Free PMC article. Updated. Review.
-
Rufinamide add-on therapy for drug-resistant epilepsy.Cochrane Database Syst Rev. 2020 Nov 8;11(11):CD011772. doi: 10.1002/14651858.CD011772.pub3. Cochrane Database Syst Rev. 2020. PMID: 33179247 Free PMC article.
Cited by
-
Improving epilepsy management by targeting P2 × 7 receptor with ROS/electric responsive nanomicelles.J Nanobiotechnology. 2025 May 5;23(1):332. doi: 10.1186/s12951-025-03386-y. J Nanobiotechnology. 2025. PMID: 40325469 Free PMC article.
References
-
- The epidemiology of drug-resistant epilepsy: a systematic review and meta-analysis. Kalilani L, Sun X, Pelgrims B, Noack-Rink M, Villanueva V. Epilepsia. 2018;59:2179–2193. - PubMed
-
- Differences in functional connectivity profiles as a predictor of response to anterior thalamic nucleus deep brain stimulation for epilepsy: a hypothesis for the mechanism of action and a potential biomarker for outcomes. Middlebrooks EH, Grewal SS, Stead M, Lundstrom BN, Worrell GA, Van Gompel JJ. Neurosurg Focus. 2018;45:0. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Research Materials
