Transcranial Direct Current Stimulation Combined With Repetitive Transcranial Magnetic Stimulation for Depression: A Randomized Clinical Trial
- PMID: 39535797
- PMCID: PMC11561687
- DOI: 10.1001/jamanetworkopen.2024.44306
Transcranial Direct Current Stimulation Combined With Repetitive Transcranial Magnetic Stimulation for Depression: A Randomized Clinical Trial
Abstract
Importance: Repetitive transcranial magnetic stimulation (rTMS) and transcranial direct current stimulation (tDCS) are both recognized as effective treatments for depression when applied individually. However, it is unknown whether rTMS combined with tDCS has better efficacy in the treatment of major depressive disorder (MDD).
Objective: To investigate the clinical effectiveness and safety of rTMS, tDCS, tDCS + rTMS, and sham tDCS + sham rTMS after 2 weeks of treatment in patients with MDD.
Design, setting, and participants: This double-blind, sham-controlled randomized clinical trial was conducted from November 2021 to April 2023 at 3 hospitals in China (Kangning Hospital affiliated with Ningbo University, Lishui Second People's Hospital, and Taizhou Second People's Hospital). Adult patients (aged 18-65 years) who were diagnosed with major depressive disorder were recruited. Participants were randomly assigned to 1 of 4 interventions: active tDCS + active rTMS, sham tDCS + active rTMS, active tDCS + sham rTMS, and sham tDCS + sham rTMS. Data analysis followed an intention-to-treat approach.
Intervention: Patients received a 2-week course of treatment. The tDCS was administered using a 2-mA direct current stimulator with electrodes placed on the left and right dorsolateral prefrontal cortex (DLPFC). Each tDCS session lasted 20 minutes and was conducted 30 to 60 minutes prior to the rTMS session for a total of 10 sessions. The rTMS was delivered at a frequency of 10 Hz using a figure-8 coil placed on the left DLPFC, with each session consisting of 1600 pulses. Treatments were administered 5 times per week for 2 weeks. Sham treatments were performed with a pseudostimulation coil and emitted only sound.
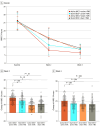
Main outcomes and measures: The primary outcome was the change in total score from baseline to week 2 on the 24-item Hamilton Depression Rating Scale (HDRS-24; score range: 0-52, with the highest score indicating more severe symptoms).
Results: A total of 240 participants (139 females [57.9%]; mean [SD] age, 32.50 [15.18] years) were included. As a primary outcome, patients who received active tDCS + active rTMS showed a significantly greater reduction in mean (SD) HDRS-24 total scores compared with patients in the other 3 groups (active tDCS + active rTMS: 18.33 [5.39], sham tDCS + active rTMS: 14.86 [5.59], active tDCS + sham rTMS: 9.21 [4.61], and sham tDCS + sham rTMS: 10.77 [5.67]; F3,236 = 35.79; η2 = 0.31 [95% CI, 0.21-0.39]; P < .001).
Conclusions and relevance: This trial found that tDCS + rTMS was a more effective and safe treatment option than either the tDCS or rTMS intervention alone for patients with MDD.
Trial registration: China Clinical Trial Registry Identifier ChiCTR2100052122.
Conflict of interest statement
Figures
References
-
- Murray CJ, Barber RM, Foreman KJ, et al. ; GBD 2013 DALYs and HALE Collaborators . Global, regional, and national disability-adjusted life years (DALYs) for 306 diseases and injuries and healthy life expectancy (HALE) for 188 countries, 1990-2013: quantifying the epidemiological transition. Lancet. 2015;386(10009):2145-2191. doi:10.1016/S0140-6736(15)61340-X - DOI - PMC - PubMed
Publication types
MeSH terms
Associated data
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

