The accuracy of ultrasensitive PSA in predicting disease progression after radical prostatectomy
- PMID: 39539557
- PMCID: PMC11557256
- DOI: 10.1002/bco2.413
The accuracy of ultrasensitive PSA in predicting disease progression after radical prostatectomy
Erratum in
-
Erratum.BJUI Compass. 2024 Dec 30;5(12):1324-1329. doi: 10.1002/bco2.482. eCollection 2024 Dec. BJUI Compass. 2024. PMID: 39744071 Free PMC article.
Abstract
Objectives: To assess the role of ultrasensitive PSA values (usPSA) after radical prostatectomy in predicting the subsequent biochemical recurrence (BCR).
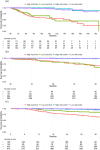
Material and methods: The study included 1836 patients who underwent open or robot-assisted RP at Turku University Hospital between 2003 and 2018. Exclusion criteria involved patients with adjuvant treatments and those who did not reach a PSA nadir <0.1 ng/ml, resulting in a final cohort of 1313 patients. The prognostic impact of the optimal usPSA nadir cut-off value 6 months after RP was investigated to predict subsequent BCR for the whole cohort (N = 1313). The optimal usPSA cut-off value was determined for patients at 3-5 years post-surgery (N = 806) and beyond 5 years (N = 493) of follow-up. We used the area under the curve (AUC) calculation and the Kaplan-Meier method.
Results: In a cohort with a median age of 64, primarily featuring Gleason score 7 prostate cancer. uPSA nadir of 0.01 ng/ml (AUC = 0.80) at the first monitoring post-surgery emerged as the optimal cut-off for identifying subjects at low (80%) or high (20%) risk of BCR within the first 3 years. Beyond this period, uPSA values during the first 3 [(AUC = 0.89; 3-5 years post-surgery) and (AUC = 0.81; beyond 5 years)] and 5 post-surgery years (AUC = 0.85) outperformed uPSA nadir in predicting subsequent BCR. Notably, EAU-defined high-risk patients with low uPSA nadir maintained substantial BCR-free survival.
Conclusion: In conclusion, a low usPSA predicts minimal BCR risk over the next 2-3 years post-measurement. Patients with low usPSA can benefit from reduced post-surgery PSA monitoring at 2- to 3-year intervals without compromising outcomes. This strategic approach optimizes resource allocation in busy urological outpatient clinics, especially valuable in publicly reimbursed healthcare systems like Finland.
Keywords: biochemical recurrence; prostate‐specific antigen; prostatic neoplasms; radical prostatectomy; ultrasensitive prostate‐specific antigen.
© 2024 The Author(s). BJUI Compass published by John Wiley & Sons Ltd on behalf of BJU International Company.
Conflict of interest statement
No potential conflict of interest was reported by the authors.
Figures

Similar articles
-
[-2]proPSA versus ultrasensitive PSA fluctuations over time in the first year from radical prostatectomy, in an high-risk prostate cancer population: A first report.BMC Urol. 2016 Mar 24;16:14. doi: 10.1186/s12894-016-0131-0. BMC Urol. 2016. PMID: 27013515 Free PMC article.
-
Detectable Prostate-specific antigen value between 0.01 and 0.1 ng/ml following robotic-assisted radical prostatectomy (RARP): does it correlate with future biochemical recurrence?World J Urol. 2021 Jun;39(6):1853-1860. doi: 10.1007/s00345-020-03367-w. Epub 2020 Jul 21. World J Urol. 2021. PMID: 32696130
-
Ultrasensitive prostate-specific antigen level as a predictor of biochemical progression after robot-assisted radical prostatectomy: Towards risk adapted follow-up.J Clin Lab Anal. 2019 Feb;33(2):e22693. doi: 10.1002/jcla.22693. Epub 2018 Oct 26. J Clin Lab Anal. 2019. PMID: 30365194 Free PMC article.
-
First Postprostatectomy Ultrasensitive Prostate-specific Antigen Predicts Survival in Patients with High-risk Prostate Cancer Pathology.Eur Urol Oncol. 2018 Oct;1(5):378-385. doi: 10.1016/j.euo.2018.07.008. Epub 2018 Aug 22. Eur Urol Oncol. 2018. PMID: 31158076
-
Ultrasensitive prostate specific antigen assay following laparoscopic radical prostatectomy--an outcome measure for defining the learning curve.Ann R Coll Surg Engl. 2009 Jul;91(5):399-403. doi: 10.1308/003588409X428289. Epub 2009 Apr 30. Ann R Coll Surg Engl. 2009. PMID: 19409146 Free PMC article.
Cited by
-
Erratum.BJUI Compass. 2024 Dec 30;5(12):1324-1329. doi: 10.1002/bco2.482. eCollection 2024 Dec. BJUI Compass. 2024. PMID: 39744071 Free PMC article.
References
-
- Mottet N, Bellmunt J, Briers E, Bolla M, Bourke L, Cornford P, et al. EAU–ESTRO–ESUR–SIOG guidelines on prostate cancer 2020. Available from: https://uroweb.org/guideline/prostate-cancer/
-
- Ferguson RA, Yu H, Kalyvas M, Zammit S, Diamandis EP. Ultrasensitive detection of prostate‐specific antigen by a time‐resolved immunofluorometric assay and the Immulite immunochemiluminescent third‐generation assay: potential applications in prostate and breast cancers. Clin. Chem. 1996;42(5):675–684. 10.1093/clinchem/42.5.675 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
