Imaging-Guided Classification of Neovascularization in Neovascular Age-Related Macular Degeneration: Progress to Date
- PMID: 39539820
- PMCID: PMC11556320
- DOI: 10.1177/24741264241276929
Imaging-Guided Classification of Neovascularization in Neovascular Age-Related Macular Degeneration: Progress to Date
Abstract
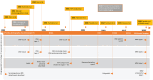
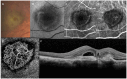
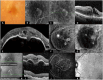
Purpose: To review the evolution of terminology describing the classification of lesions in neovascular age-related macular degeneration (nAMD) based on retinal imaging technologies. Methods: A review of the current and historical literature on imaging-guided classification of neovascularization in nAMD was performed. Results: Imaging-guided classification of neovascularization in nAMD facilitates an understanding of the pathological mechanisms and disease progression. Neovascularization classification has evolved with advances in imaging technologies, from earlier classifications based on neovascularization patterns assessed by fluorescein angiography to multimodal imaging patterns, resulting in varied descriptions of lesions depending on the techniques used. Until recently, there has been a lack of consensus regarding the clinical features of choroidal neovascularization lesion types as a result of the imaging modalities initially used to define them; a recent consensus on classification has the potential to simplify and clarify descriptions of neovascularization in nAMD. The use of multimodal imaging techniques will improve lesion identification and has the potential to individualize treatment plans and improve outcomes. Conclusions: Widespread adoption of a consensus-based, image-guided classification system for neovascular lesions in nAMD and the appropriate imaging techniques used to identify them will aid clinical research and could potentially improve patient outcomes by individualizing treatment plans in the future.
Keywords: age-related macular degeneration; choroidal neovascularization; fluorescein angiography; imaging; indocyanine green angiography; macular neovascularization; multimodal imaging; neovascularization; optical coherence tomography angiography.
© The Author(s) 2024.
Conflict of interest statement
Dr. Wolf received funds from Bayer, Novartis, Sandoz, and Zeiss. Dr. Chow received consultancy fees from Bayer and Roche, research support from Opthea and RegenxBio Inc, and payment or honoraria for lectures or educational activities from Aviceda Therapeutics and Apellis Pharmaceuticals. Dr. Souied provided expert support for AbbVie, Apellis Pharmaceuticals, Bayer, Novartis, Roche, and Teva. Dr. Viola received consultancy fees, grants, and support for attending meetings and/or travel and participation on a data safety monitoring board or advisory board from Bayer, Novartis, and Roche. Dr. Kaiser received consultancy fees from Alcon, Allergan, Bayer, Bausch + Lomb, Biogen Idec, Clearside Biomedical, Coherus BioSciences, Genentech/Roche, Astellas, Novartis, Ocular Therapeutix, Oculis, Ocuphire Pharma, Regeneron Pharmaceuticals Inc, and RegenxBio Inc. Dr. Staurenghi received consultant/advisor fees from AbbVie, Annexon Bioscience, Apellis Pharmaceuticals, Bayer HealthCare Pharmaceutical, Boehringer Ingelheim, Genentech, Iveric Bio, and OraPharma Inc; consultant/advisor fees and grant support from Optos Inc and RetinAI; grant support from Quantel Medical; consultant/advisor fees, lecture fees/speakers bureau and grant support from CenterVue, Inc, Heidelberg Engineering, Hoffman La Roche, and Novartis Pharmaceuticals; lecture fees/speakers bureau and grant support from Carl Zeiss Meditec; consultant/advisor fees and lecture fees/speakers bureau from Medscape; lecture fees/speakers bureau and grant support from Nidek Inc; and patents/royalty from Ocular Instruments Inc. Dr. Gallego-Pinazo received consultancy fees from Apellis Pharmaceuticals and Carl Zeiss AG, consultancy fees and research support from Novartis and Roche, and research support from Ionis, Iveric Bio, Janssen, and Opthea. Dr. Iida received consultancy fees from Bayer Yakuhin Ltd., Chugai Pharmaceutical Co, Janssen Pharmaceutical K.K., Kyowa Kirin, Nippon Boehringer Ingelheim Co, Ltd, Novartis, and Senju Pharmaceutical Co, Ltd; study funding and article processing charges from Bayer AG (Leverkusen, Germany); funding for editorial support and medical writing from Bayer Consumer Care AG (Basel, Switzerland); study funding from Regeneron Pharmaceuticals Inc; grant funding from Alcon Japan, AMO Pharma Ltd, HOYA, Nidek, Novartis, Santen Pharmaceutical, Senju Pharmaceutical Co Ltd, and Topcon Healthcare; payment or honoraria for lectures from Alcon Japan, Bayer Yakuhin Ltd, Canon Inc, Chugai Pharmaceutical Co, Nidek, Nikon, Novartis, Otsuka Pharmaceutical Co, Ltd, Santen Pharmaceutical, Senju Pharmaceutical Co, Ltd; and Topcon; patent from Topcon; and other financial rewards from Kyowa Kirin.
Figures



References
-
- Spaide RF, Jaffe GJ, Sarraf D, et al. Consensus nomenclature for reporting neovascular age-related macular degeneration data: Consensus on Neovascular Age-Related Macular Degeneration Nomenclature study group. Ophthalmology. 2020;127(5):616-636. doi: 10.1016/j.ophtha.2019.11.004 - DOI - PMC - PubMed
-
- Bressler NM, Treatment of Age-Related Macular Degeneration with Photodynamic Therapy (TAP) Study Group. Photodynamic therapy of subfoveal choroidal neovascularization in age-related macular degeneration with verteporfin: two-year results of 2 randomized clinical trials—TAP report 2. Arch Ophthalmol. 2001;119(2):198-207. - PubMed
-
- Photodynamic therapy of subfoveal choroidal neovascularization in age-related macular degeneration with verteporfin: one-year results of 2 randomized clinical trials—TAP report. Treatment of Age-Related Macular Degeneration with Photodynamic Therapy (TAP) Study Group. Arch Ophthalmol. 1999;117(10):1329-1345. - PubMed

