Nivolumab and sunitinib in patients with advanced bone sarcomas: A multicenter, single-arm, phase 2 trial
- PMID: 39540661
- PMCID: PMC11694334
- DOI: 10.1002/cncr.35628
Nivolumab and sunitinib in patients with advanced bone sarcomas: A multicenter, single-arm, phase 2 trial
Abstract
Background: Herein, we present the results of the phase 2 IMMUNOSARC study (NCT03277924), investigating sunitinib and nivolumab in adult patients with advanced bone sarcomas (BS).
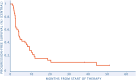
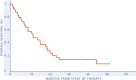
Methods: Progressing patients with a diagnosis of BS were eligible. Treatment was comprised of sunitinib (37.5 mg/day on days 1-14, 25 mg/day afterword) plus nivolumab (3 mg/kg every 2 weeks). Primary end point was progression-free survival rate (PFSR) at 6 months based on central radiology review. Secondary end points were overall survival (OS), overall response rate (ORR) by Response Evaluation Criteria in Solid Tumors (RECIST) v1.1, and safety.
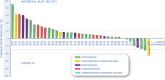
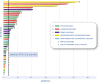
Results: A total of 46 patients were screened, 40 patients entered the study, and 38 underwent central radiological review and were evaluable for primary end point. Median age was 47 years (range, 21-74). Histologies include 17 (43%) osteosarcoma, 14 chondrosarcoma (35%, 10 conventional, four dedifferentiated [DDCS]), eight (20%) Ewing sarcoma, and one (2%) undifferentiated pleomorphic sarcoma. The PFSR at 6 months was 42% (95% confidence interval [CI], 27-58). With a median follow-up of 39.8 months (95% CI, 37.9-41.7), the median PFS and OS were 3.8 months (95% CI, 2.7-4.8) and 11.9 months (95% CI, 5.6-18.2). ORR by RECIST was 5%, with two of 38 partial responses (one of four DDCS and one of 17 osteosarcoma), 19 of 38 (50%) stable disease, and 17 of 38 (45%) progressions. Grade ≥3 adverse events were neutropenia (six of 40, 15%), anemia (5/40, hypertension (6/40, 15%), 12.5%), ALT/AST elevation (5/40, 12.5%), and pneumonitis (1/40, 2.5%). Seventeen percent of patients discontinued treatment due to toxicity, including a treatment-related grade 5 pneumonitis CONCLUSION: The trial met its primary end point in the BS cohort with >15% of patients progression-free at 6 months. However, the toxicity profile of this regimen was relevant.
Keywords: Ewing sarcoma; PD‐L1 inhibitor; anti‐angiogeninic; bone; dedifferentiated chondrosarcoma; immunotherapy; nivolumab; osteosarcoma; sarcoma; sunitinib.
© 2024 The Author(s). Cancer published by Wiley Periodicals LLC on behalf of American Cancer Society.
Conflict of interest statement
Emanuela Palmerini has served on advisory boards for Daiichi Sankyo, Deciphera Pharmaceuticals, Eusa Pharma, and SynOx Therapeutics. Giovanni Grignani reports grants and personal fees from PharmaMar, grants from Novartis, and personal consulting fees from Lilly, Pfizer, Bayer, and Eisai. Andres Redondo reports grants and personal fees from PharmaMar; personal fees from Lilly, Novartis, Amgen, AstraZeneca, and Tesaro; grants and personal fees from Roche; and grants from Eisai. Nadia Hindi reports grants, personal fees, and nonfinancial support from PharmaMar; personal fees from Lilly; grants from Eisai and Novartis; and research funding for clinical studies (institutional) from PharmaMar, Eli Lilly, AROG, Bayer, Eisai, Lixte, Karyopharm, Deciphera, GlaxoSmithKline, Novartis, Blueprint, Nektar, Forma, Amgen, and Daiichi Sankyo. Salvatore Provenzano reports personal fees for consultancy from Italfarmaco and Boehringer Ingelheim; and the spouse employed in AstraZeneca. Silvia Stacchiotti reports personal financial interests including honoraria, consultancy, or advisory role from Agentus, Bayer, Boehringer Ingelheim, Daiichi Sankyo, Deciphera, Ikena, Gentili, GlaxoSmithKline, Ipsen, Nec Oncolimmunity, Novartis, Pharmamar, Pharma Essentia, Regeneron Springworks, and Servier; and institutional financial interests from Abbisko, Adaptimmune, Advenchen, Bayer, Blueprint, Boehringer Ingelheim, Daiichi Sankyo, Deciphera, EISAI, Epizyme, Foghorn, Hutchinson, Inhbrix, Karyopharm, Novartis, Pharmamar, RainThera, and Springworks. Jose Antonio Lopez Martin reports honoraria for advisory board participation and travel support from PharmaMar, Eli Lilly, Bayer, Eisai, Novartis, Bristol‐Myers Squibb, MSD, Roche, Celgene, Pierre Fabre, Pfizer, GlaxoSmithKline, Daiichi Sankyo, Amgen, and Chobani. Javier Martinez Trufero reports honoraria for advisory board participation and travel support from PharmaMar, Eli Lilly, Eisai, Merck Sharp & Dohm, Merck, GlaxoSmithKline, and Roche. Lorenzo D’Ambrosio reports advisory board participation for Boehringer Ingelheim, AstraZeneca, PSI CRO Italy, and GlaxoSmithKline; and travel support from PharmaMar, GlaxoSmithKline, and AstraZeneca. Enrique de Alava reports personal fees and nonfinancial support from Roche, Bristol‐Myers Squibb, and PharmaMar; and personal fees from Bayer. David S. Moura reports institutional research grants from PharmaMar, Eisai, Immix BioPharma, and Novartis outside the submitted work; travel support from PharmaMar, Eisai, Celgene, Bayer, and Pfizer, and personal fees from Tecnopharma. Javier Martin Broto reports research grants from PharmaMar, Eisai, Immix BioPharma, and Novartis; honoraria for advisory board participation and expert testimony from PharmaMar, Eli Lilly, Bayer, and Eisai; and research funding for clinical studies (institutional) from PharmaMar, Eli Lilly, AROG, Bayer, Eisai, Lixte, Karyopharm, Deciphera, GlaxoSmithKline, Novartis, Blueprint, Nektar, Forma, Amgen, and Daiichi Sankyo. The other authors declare no conflicts of interest.
Figures

References
-
- McCabe M, Kirton L, Khan M, et al. Phase III assessment of topotecan and cyclophosphamide and high‐dose ifosfamide in rEECur: an international randomized controlled trial of chemotherapy for the treatment of recurrent and primary refractory Ewing sarcoma (RR‐ES). J Clin Oncol. 2022;40(suppl 17):LBA2. doi:10.1200/jco.2022.40.17_suppl.lba2 - DOI
-
- van Ewijk R, Cleirec M, Herold N, et al. FOSTER Consortium (Fight OsteoSarcoma Through European Research), work package 3 on recurrent/refractory osteosarcoma trials. A systematic review of recent phase‐II trials in refractory or recurrent osteosarcoma: can we inform future trial design? Cancer Treat Rev. 2023;120:102625. doi:10.1016/j.ctrv.2023.102625 - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

