Discovery and Preclinical Characterization of Fulacimstat (BAY 1142524), a Potent and Selective Chymase Inhibitor As a New Profibrinolytic Approach for Safe Thrombus Resolution
- PMID: 39541507
- PMCID: PMC11956016
- DOI: 10.1021/acs.jmedchem.4c01819
Discovery and Preclinical Characterization of Fulacimstat (BAY 1142524), a Potent and Selective Chymase Inhibitor As a New Profibrinolytic Approach for Safe Thrombus Resolution
Abstract
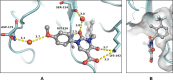
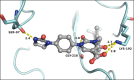
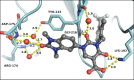
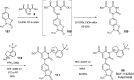
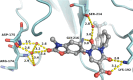
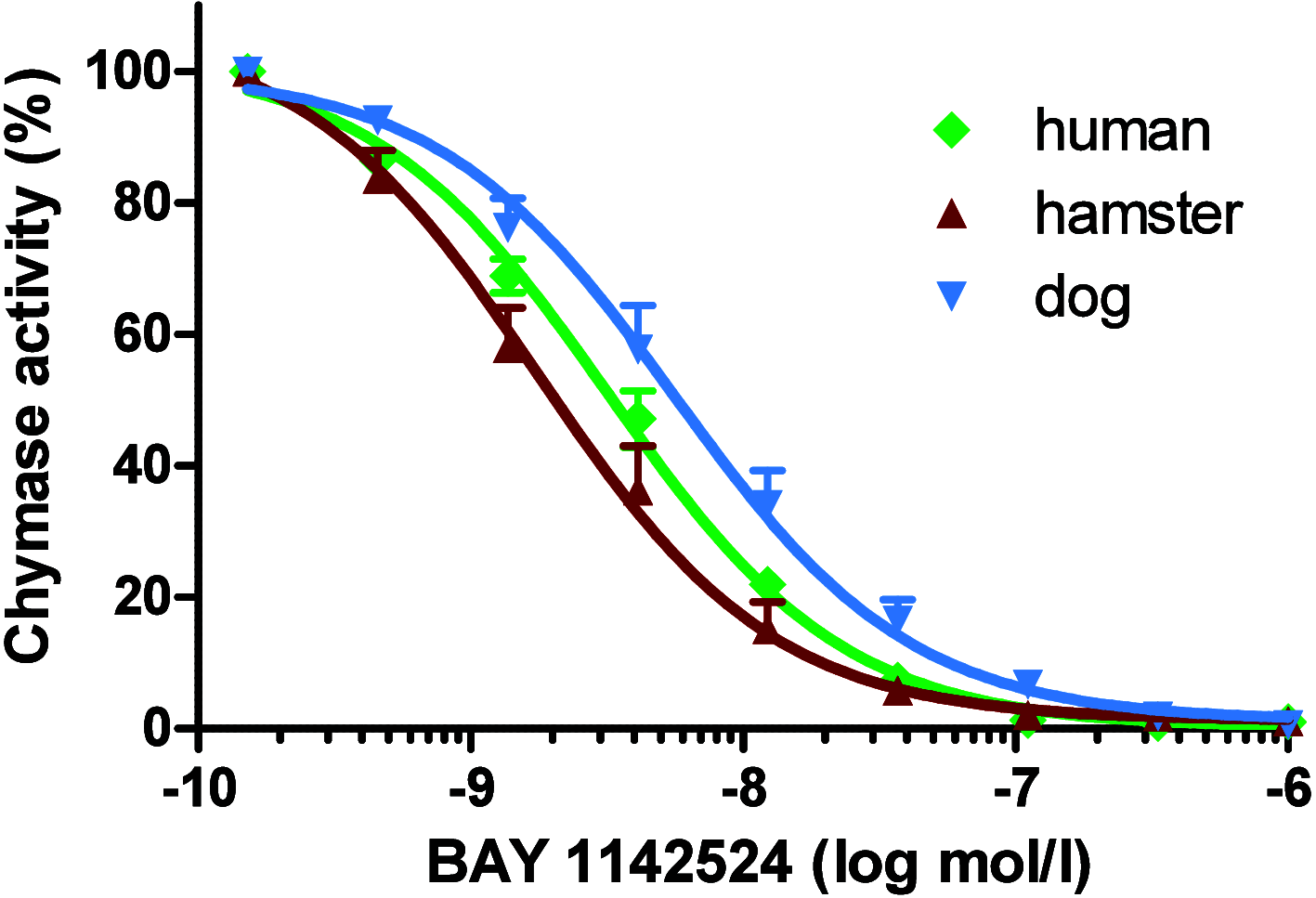
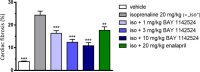
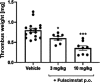
Chymase is a serine-protease produced by mast cells. In the past few decades, its role in fibrotic diseases triggered the search for orally available chymase inhibitors. Aiming at reducing adverse cardiac remodeling after myocardial infarction, our research efforts resulted in the discovery of fulacimstat (BAY 1142524). While clinical trials did not demonstrate efficacy in this indication, the recent discovery of a new unexpected biological role of chymase spurred a revival of interest in chymase inhibition: chymase was shown to inactivate plasmin within fibrin-rich clots. Chymase inhibitors are now considered as potential profibrinolytic drugs with low bleeding risk and therefore exceptional safety for the treatment of acute thrombosis settings such as stroke, pulmonary embolism, or venous thrombosis. This article describes the chemical optimization journey from a screening hit to the discovery of fulacimstat (BAY 1142524), a selective chymase inhibitor with a good safety profile, as well as its preclinical in vitro and in vivo characterization.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
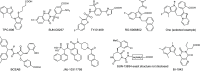
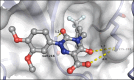
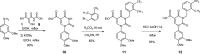
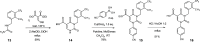

Similar articles
-
Recombinant chymase inhibits fibrinolysis induced by endogenous plasmin in clotted human blood.Front Immunol. 2025 Apr 17;16:1511990. doi: 10.3389/fimmu.2025.1511990. eCollection 2025. Front Immunol. 2025. PMID: 40313964 Free PMC article.
-
Opening the Door to a New Treatment Paradigm: The Re-emergence of Chymase Inhibitors.J Med Chem. 2025 Mar 27;68(6):6100-6103. doi: 10.1021/acs.jmedchem.5c00413. Epub 2025 Feb 17. J Med Chem. 2025. PMID: 39961794 Review.
-
Effects of the chymase inhibitor fulacimstat on adverse cardiac remodeling after acute myocardial infarction-Results of the Chymase Inhibitor in Adverse Remodeling after Myocardial Infarction (CHIARA MIA) 2 trial.Am Heart J. 2020 Jun;224:129-137. doi: 10.1016/j.ahj.2020.01.012. Epub 2020 Jan 25. Am Heart J. 2020. PMID: 32375104 Clinical Trial.
-
Safety and Tolerability of the Chymase Inhibitor Fulacimstat in Patients With Left Ventricular Dysfunction After Myocardial Infarction-Results of the CHIARA MIA 1 Trial.Clin Pharmacol Drug Dev. 2019 Oct;8(7):942-951. doi: 10.1002/cpdd.633. Epub 2018 Nov 19. Clin Pharmacol Drug Dev. 2019. PMID: 30452784 Clinical Trial.
-
Pathological roles of angiotensin II produced by mast cell chymase and the effects of chymase inhibition in animal models.Pharmacol Ther. 2006 Dec;112(3):668-76. doi: 10.1016/j.pharmthera.2006.05.008. Epub 2006 Jul 11. Pharmacol Ther. 2006. PMID: 16837049 Review.
References
-
- Caughey G. H. Mast cell tryptases and chymases in inflammation and host defense. Immunological Reviews 2007, 217, 141–154. 10.1111/j.1600-065X.2007.00509.x. - DOI - PMC - PubMed
- Shiota N.; Rysä J.; Kovanen P. T.; Ruskoaho H.; Kokkonen J. O.; Lindstedt K. A. A role for cardiac mast cells in the pathogenesis of hypertensive heart disease. J. Hypertens. 2003, 21 (10), 1935–1944. 10.1097/00004872-200310000-00022. - DOI - PubMed
- Pejler G. Novel insight into the in vivo function of mast cell chymase: lessons from knockouts and inhibitors. J. Innate Immun. 2020, 12 (5), 357–372. 10.1159/000506985. - DOI - PMC - PubMed
- Hellman L.; Akula S.; Fu Z.; Wernersson S. Mast cells and basophil granule proteases - in vivo targets and function. Frontiers in Immunology 2022, 13, 918305.10.3389/fimmu.2022.918305. - DOI - PMC - PubMed
-
- Kovanen P. T.; Kaartinen M.; Paavonen T. Infiltrates of activated mast cells at the site of coronary atheromatous erosion or rupture in myocardial infarction. Circulation 1995, 92 (5), 1084–1088. 10.1161/01.CIR.92.5.1084. - DOI - PubMed
- Sun J.; Zhang J.; Lindholt J. S.; Sukhova G. K.; Liu J.; He A.; Abrink M.; Pejler G.; Stevens R. L.; Thompson R. W.; Ennis T. L.; Gurish M. F.; Libby P.; Shi G. P. Critical role of mast cell chymase in mouse abdominal aortic aneurysm formation. Circulation 2009, 120 (11), 973–982. 10.1161/CIRCULATIONAHA.109.849679. - DOI - PMC - PubMed
- Bacani C.; Frishman W. H. Chymase, a new pharmacologic target in cardiovascular disease. Cardiol. Rev. 2006, 14 (4), 187–193. 10.1097/01.crd.0000195220.62533.c5. - DOI - PubMed
-
- Huang X. R.; Chen W. Y.; Truong L. D.; Lan H. Y. Chymase is upregulated in diabetic nephropathy: implications for an alternative pathway of angiotensin II-mediated diabetic renal and vascular disease. J. Am. Soc. Nephrol. 2003, 14 (7), 1738–1747. 10.1097/01.ASN.0000071512.93927.4E. - DOI - PubMed
- McPherson E. A.; Luo Z.; Brown R. A.; LeBard L. S.; Corless C. C.; Speth R. C.; Bagby S. P. Chymase-like angiotensin II-generating activity in end-stage human autosomal dominant polycystic kidney disease. J. Am. Soc. Nephrol. 2004, 15 (2), 493–500. 10.1097/01.ASN.0000109782.28991.26. - DOI - PubMed
- Maeda Y.; Inoguchi T.; Takei R.; Sawada F.; Sasaki S.; Fujii M.; Kobayashi K.; Urata H.; Nishiyama A.; Takayanagi R. Inhibition of chymase protects against diabetes-induced oxidative stress and renal dysfunction in hamsters. Am. J. Physiol. Renal Physiol. 2010, 299 (6), F1328–F1338. 10.1152/ajprenal.00337.2010. - DOI - PubMed
-
- Doggrell S. A Therapeutic potential of non-peptide chymase inhibitors. Expert Opin. Ther. Patents 2008, 18, 485–499. and references cited therein,10.1517/13543776.18.5.485. - DOI
- Hirata K.; Sugama Y.; Ikura Y.; Ohsawa M.; Inoue Y.; Yamamoto S.; Kitaichi M.; Ueda M. Enhanced mast cell chymase expression in human idiopathic interstitial pneumonia. Int. J. Mol. Med. 2007, 19 (4), 565–570. 10.3892/ijmm.19.4.565. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

