A non-canonical role of somatic Cyclin D/CYD-1 in oogenesis and in maintenance of reproductive fidelity, dependent on the FOXO/DAF-16 activation state
- PMID: 39546504
- PMCID: PMC11602045
- DOI: 10.1371/journal.pgen.1011453
A non-canonical role of somatic Cyclin D/CYD-1 in oogenesis and in maintenance of reproductive fidelity, dependent on the FOXO/DAF-16 activation state
Abstract
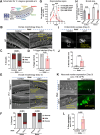
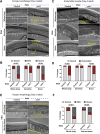
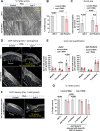
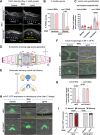
For the optimal survival of a species, an organism coordinates its reproductive decisions with the nutrient availability of its niche. Thus, nutrient-sensing pathways like insulin-IGF-1 signaling (IIS) play an important role in modulating cell division, oogenesis, and reproductive aging. Lowering of the IIS leads to the activation of the downstream FOXO transcription factor (TF) DAF-16 in Caenorhabditis elegans which promotes oocyte quality and delays reproductive aging. However, less is known about how the IIS axis responds to changes in cell cycle proteins, particularly in the somatic tissues. Here, we show a new aspect of the regulation of the germline by this nutrient-sensing axis. First, we show that the canonical G1-S cyclin, Cyclin D/CYD-1, regulates reproductive fidelity from the uterine tissue of wild-type worms. Then, we show that knocking down cyd-1 in the uterine tissue of an IIS receptor mutant arrests oogenesis at the pachytene stage of meiosis-1 in a DAF-16-dependent manner. We observe activated DAF-16-dependent deterioration of the somatic gonadal tissues like the sheath cells, and transcriptional de-regulation of the sperm-to-oocyte switch genes which may be the underlying reason for the absence of oogenesis. Deleting DAF-16 releases the arrest and leads to restoration of the somatic gonad but poor-quality oocytes are produced. Together, our study reveals the unrecognized cell non-autonomous interaction of Cyclin D/CYD-1 and FOXO/DAF-16 in the regulation of oogenesis and reproductive fidelity.
Copyright: © 2024 Rautela et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


Similar articles
-
DNA damage signals from somatic uterine tissue arrest oogenesis through activated DAF-16.Development. 2023 Sep 1;150(17):dev201472. doi: 10.1242/dev.201472. Epub 2023 Sep 12. Development. 2023. PMID: 37577954
-
Insulin Signaling Regulates Oocyte Quality Maintenance with Age via Cathepsin B Activity.Curr Biol. 2018 Mar 5;28(5):753-760.e4. doi: 10.1016/j.cub.2018.01.052. Epub 2018 Feb 22. Curr Biol. 2018. PMID: 29478855 Free PMC article.
-
The C. elegans adult neuronal IIS/FOXO transcriptome reveals adult phenotype regulators.Nature. 2016 Jan 7;529(7584):92-6. doi: 10.1038/nature16483. Epub 2015 Dec 14. Nature. 2016. PMID: 26675724 Free PMC article.
-
DAF-16/FoxO in Caenorhabditis elegans and Its Role in Metabolic Remodeling.Cells. 2020 Jan 2;9(1):109. doi: 10.3390/cells9010109. Cells. 2020. PMID: 31906434 Free PMC article. Review.
-
DAF-16: FOXO in the Context of C. elegans.Curr Top Dev Biol. 2018;127:1-21. doi: 10.1016/bs.ctdb.2017.11.007. Epub 2018 Feb 2. Curr Top Dev Biol. 2018. PMID: 29433733 Review.
References
-
- Sasaki H, Hamatani T, Kamijo S, Iwai M, Kobanawa M, Ogawa S, et al.. Impact of Oxidative Stress on Age-Associated Decline in Oocyte Developmental Competence. Front Endocrinol (Lausanne). 2019;10:811. Epub 20191122. doi: 10.3389/fendo.2019.00811 ; PubMed Central PMCID: PMC6882737. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

