Protocol for Therapeutic Drug Monitoring Within the Clinical Range Using Mid-infrared Spectroscopy
- PMID: 39557616
- PMCID: PMC11618749
- DOI: 10.1021/acs.analchem.4c03864
Protocol for Therapeutic Drug Monitoring Within the Clinical Range Using Mid-infrared Spectroscopy
Abstract
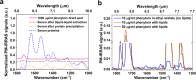
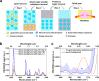
Therapeutic drug monitoring (TDM), which involves measuring drug levels in patients' body fluids, is an important procedure in clinical practice. However, the analysis technique currently used, i.e. liquid chromatography-tandem mass spectrometry (LC-MS/MS), is laboratory-based, so does not offer the short response time that is often required by clinicians. We suggest that techniques based on Fourier transform infrared spectroscopy (FTIR) offer a promising alternative for TDM. FTIR is rapid, highly specific and can be miniaturized for near-patient applications. The challenge, however, is that FTIR for TDM is limited by the strong mid-IR absorption of endogenous serum constituents. Here, we address this issue and introduce a versatile approach for removing the background of serum lipids, proteins and small water-soluble substances. Using phenytoin, an antiepileptic drug, as an example, we show that our approach enables FTIR to precisely quantify drug molecules in human serum at clinically relevant levels (10 μg/mL), providing an efficient analysis method for TDM. Beyond mid-IR spectroscopy, our study is applicable to other drug sensing techniques that suffer from the large background of serum samples.
Conflict of interest statement
The authors declare no competing financial interest.
Figures


Similar articles
-
Therapeutic Drug Monitoring of Phenytoin by Simple, Rapid, Accurate, Highly Sensitive and Novel Method and Its Clinical Applications.Curr Pharm Biotechnol. 2017;18(13):1098-1105. doi: 10.2174/1389201019666180209164444. Curr Pharm Biotechnol. 2017. PMID: 29437004
-
Quantification of Free Phenytoin by Liquid Chromatography Tandem Mass Spectrometry (LC/MS/MS).Methods Mol Biol. 2016;1383:241-6. doi: 10.1007/978-1-4939-3252-8_25. Methods Mol Biol. 2016. PMID: 26660192
-
Detection of the antiepileptic drug phenytoin using a single free-standing piezoresistive microcantilever for therapeutic drug monitoring.Biosens Bioelectron. 2014 Sep 15;59:233-8. doi: 10.1016/j.bios.2014.03.047. Epub 2014 Mar 28. Biosens Bioelectron. 2014. PMID: 24732600
-
Applications of mid-infrared spectroscopy in the clinical laboratory setting.Crit Rev Clin Lab Sci. 2018 Jan;55(1):1-20. doi: 10.1080/10408363.2017.1414142. Epub 2017 Dec 14. Crit Rev Clin Lab Sci. 2018. PMID: 29239240 Review.
-
Screening of synthetic PDE-5 inhibitors and their analogues as adulterants: analytical techniques and challenges.J Pharm Biomed Anal. 2014 Jan;87:176-90. doi: 10.1016/j.jpba.2013.04.037. Epub 2013 May 6. J Pharm Biomed Anal. 2014. PMID: 23721687 Review.
References
-
- Rybak M.; Lomaestro B.; Rotschafer J. C.; Moellering R. Jr.; Craig W.; Billeter M.; Dalovisio J. R.; Levine D. P. Therapeutic monitoring of vancomycin in adult patients: A consensus review of the American Society of Health-System Pharmacists, the Infectious Diseases Society of America, and the Society of Infectious Diseases Pharmacists. Am. J. Health Syst. Pharm. 2009, 66 (1), 82–98. 10.2146/ajhp080434. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

