IFN-γ-induced Th1-Treg polarization in inflamed brains limits exacerbation of experimental autoimmune encephalomyelitis
- PMID: 39560646
- PMCID: PMC11621829
- DOI: 10.1073/pnas.2401692121
IFN-γ-induced Th1-Treg polarization in inflamed brains limits exacerbation of experimental autoimmune encephalomyelitis
Abstract
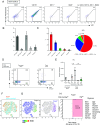
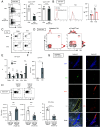
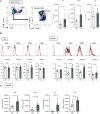
Experimental autoimmune encephalomyelitis (EAE) is the most widely used rodent model for multiple sclerosis. Interferon-γ (IFN-γ) and regulatory T cells (Tregs) are individually well known to play beneficial roles in amelioration of EAE. However, little is known about the relationship between IFN-γ and Tregs during the disease. Here, we show that IFN-γ polarizes Tregs into T helper 1 (Th1)-type Tregs (Th1-Tregs) to recover from EAE. Single-cell RNA sequencing analysis revealed that brain Tregs showed signs of IFN-γ stimulation during EAE. Loss of IFN-γ signaling in Tregs and of T cell-derived IFN-γ impaired the Th1-Treg polarization and worsened the disease. Moreover, selective ablation of Th1-Tregs using an intersectional genetic method promoted proinflammatory features of macrophages in the inflamed brains and exacerbated the EAE. Taken together, our study highlights a critical role of T cell-derived IFN-γ for Th1-Treg polarization in inflamed brain to ameliorate EAE.
Keywords: EAE; IFN-γ; Th1-Treg; VeDTR.
Conflict of interest statement
Competing interests statement:The authors declare no competing interest.
Figures




References
-
- Dhaiban S., et al. , Role of peripheral immune cells in multiple sclerosis and experimental autoimmune encephalomyelitis. Sci 3, 12 (2021).
-
- Olsson T., Critical influences of the cytokine orchestration on the outcome of myelin antigen-specific T-cell autoimmunity in experimental autoimmune encephalomyelitis and multiple sclerosis. Immunol. Rev. 144, 245–268 (1995). - PubMed
-
- Billiau A., et al. , Enhancement of experimental allergic encephalomyelitis in mice by antibodies against IFN-gamma. J. Immunol. 140, 1506–1510 (1988). - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

