Laterality, sexual dimorphism, and human vagal projectome heterogeneity shape neuromodulation to vagus nerve stimulation
- PMID: 39562711
- PMCID: PMC11576867
- DOI: 10.1038/s42003-024-07222-1
Laterality, sexual dimorphism, and human vagal projectome heterogeneity shape neuromodulation to vagus nerve stimulation
Abstract
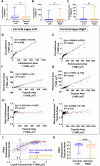
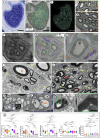
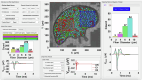
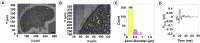
Neuromodulation by vagus nerve stimulation (VNS) provides therapeutic benefits in multiple medical conditions, including epilepsy and clinical depression, but underlying mechanisms of action are not well understood. Cervical vagus nerve biopsies were procured from transplant organ donors for high resolution light microscopy (LM) and transmission electron microscopy (TEM) to map the human fascicular and sub-fascicular organization. Cervical vagal segments show laterality with right sided dominance in fascicle numbers and cross-sectional areas as well as sexual dimorphism with female dominance in fascicle numbers. The novel and unprecedented detection of numerous small fascicles by high resolution LM and TEM expand the known fascicle size range and morphological diversity of the human vagus nerve. Ground truth TEM quantification of all myelinated and unmyelinated axons within individual nerve fascicles show marked sub-fascicular heterogeneity of nerve fiber numbers, size, and myelination. A heuristic action potential interpreter (HAPI) tool predicts VNS-evoked compound nerve action potentials (CNAPs) generated by myelinated and unmyelinated nerve fibers and validates functional dissimilarity between fascicles. Our findings of laterality, sexual dimorphism, and an expanded range of fascicle size heterogeneity provide mechanistic insights into the varied therapeutic responses and off-target effects to VNS and may guide new refinement strategies for neuromodulation.
© 2024. This is a U.S. Government work and not under copyright protection in the US; foreign copyright protection may apply.
Conflict of interest statement
Figures





References
-
- Morris, G. L. 3rd & Mueller, W. M. Long-term treatment with vagus nerve stimulation in patients with refractory epilepsy. The Vagus Nerve Stimulation Study Group E01-E05. Neurology53, 1731–1735 (1999). - PubMed
-
- Elger, G., Hoppe, C., Falkai, P., Rush, A. J. & Elger, C. E. Vagus nerve stimulation is associated with mood improvements in epilepsy patients. Epilepsy Res.42, 203–210 (2000). - PubMed
-
- Harden, C. L. et al. A Pilot study of mood in epilepsy patients treated with vagus nerve stimulation. Epilepsy Behav.1, 93–99 (2000). - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources

