Trends in medications for autoimmune disorders during pregnancy and factors for their discontinuation: a population-based study
- PMID: 39563243
- PMCID: PMC11575194
- DOI: 10.1186/s12884-024-06932-y
Trends in medications for autoimmune disorders during pregnancy and factors for their discontinuation: a population-based study
Abstract
Objectives: The medications used for autoimmune diseases have significantly evolved in recent years, but there is limited knowledge about how treatment practices changed during pregnancy. This study aimed to describe the temporal trends of immunosuppressants, immunomodulators and biologics use during pregnancy among women with autoimmune diseases, compare their use before, during, and after pregnancy, and identify factors predicting the discontinuation of these medications during pregnancy.
Methods: Using data from the Quebec Pregnancy Cohort (1998-2015), which included women under the RAMQ prescription drug plan for at least 12 months before and after pregnancy, the analysis focused on those with at least one International Classification of Diseases Ninth or Tenth Revision code in the year before pregnancy for inflammatory bowel disease, rheumatoid arthritis, spondylarthropathies, connective tissue diseases, systemic lupus erythematosus, or vasculitis. Exposure to immunosuppressants, immunomodulators and biologics were evaluated before and during the pregnancy. Discontinuation during pregnancy was defined as having no prescriptions filled during pregnancy or overlapping with the first day of gestation (1DG), given that at least one prescription was filled in the year prior to pregnancy. Generalized estimating equations were applied to estimate adjusted odds ratios (aOR) for predicting medication discontinuation during pregnancy.
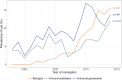
Results: Among 441,570 pregnant women, 3,285 had autoimmune diseases. From 1998 to 2014, the use of immunomodulators increased from 3.7% to 11.9%, immunosuppressants from 4.1% to 13.7%, and biologics from 0% to 15.6%. During pregnancy, compared to before, there was a significant decrease in exposure to immunomodulators (8.6% to 5.4%), immunosuppressants (14.2% to 8.7%), and biologics (5.1% to 4.7%). Factors influencing discontinuation varied by medication type; for immunosuppressants, prior biologics use (aOR = 2.12, 95%CI 1.16-3.85) and the year of pregnancy (aOR = 0.93, 95%CI 0.89-0.98) were key factors, while for biologics, it was only the year of pregnancy (aOR = 0.68, 95%CI 0.54-0.86).
Conclusions: The use of immunomodulators, immunosuppressants, and biologics has increased over time. However, exposure during pregnancy decreased, with recent years showing a lower rate of discontinuation. Understanding the factors influencing medication discontinuation during pregnancy can improve management strategies for women with autoimmune diseases.
Keywords: Autoimmune disease; Biologics; Inflammatory bowel disease; Pregnancy; Rheumatoid arthritis; Spondyloarthropathies.
© 2024. The Author(s).
Conflict of interest statement
Figures



References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

