Human studies of the efficacy and safety of stem cells in the treatment of diabetic peripheral neuropathy: a systematic review and meta-analysis
- PMID: 39563393
- PMCID: PMC11577959
- DOI: 10.1186/s13287-024-04033-3
Human studies of the efficacy and safety of stem cells in the treatment of diabetic peripheral neuropathy: a systematic review and meta-analysis
Abstract
Objective: To assess the efficacy and safety of stem cell therapy in human studies for diabetic peripheral neuropathy (DPN).
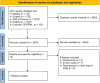
Methods: A comprehensive literature review was performed across multiple databases, including Ovid MEDLINE ALL, Embase via Ovid SP, Scopus, Web of Science Core Collection, and Cochrane CENTRAL, up to January 31, 2024. Keywords and controlled vocabularies related to diabetic neuropathy and stem cell therapy were used. Inclusion criteria encompassed all controlled trials examining stem cell therapy for DPN, excluding animal or in vitro studies, review papers, conference abstracts, and editor letters. Data extraction and risk of bias assessment were independently performed by multiple reviewers using standardized tools.
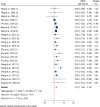
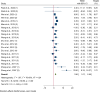
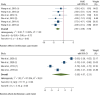
Results: Out of 5431 initial entries, seven were included. Stem cell therapies included bone marrow-derived mononuclear cells and umbilical cord-derived mesenchymal stem cells, administered mainly via intramuscular transplantation. Meta-analysis indicated significant improvements in motor nerve conduction velocity (weighted mean differences (WMD): 2.2, 95% CI 1.6-2.8) and sensory nerve conduction velocity (WMD: 1.9, 95% CI 1.1-2.6). Vibration perception threshold and Toronto Clinical Scoring System scores decreased significantly (WMD: - 2.9, 95% CI - 4.0, - 1.8, and WMD: - 3.6, 95% CI - 5.0, - 2.2, respectively). Sensitivity analysis and subgroup analysis confirmed the robustness and specificity of these findings. The complications were pain and swelling at the injection sites, which disappeared in a few days.
Conclusion: Stem cell therapy shows significant promise in improving clinical outcomes for DPN, with evident benefits in nerve conduction and sensory parameters. Further research is needed to consolidate these findings and optimize therapeutic protocols.
Keywords: Adult stem cell; Bone marrow cell transplantation; Diabetic neuropathy; Meta-analysis; Umbilical cord blood stem cell transplantation.
© 2024. The Author(s).
Conflict of interest statement
Figures

References
-
- He D, Xu Y, Xiong X, Yin C, Lei S, Cheng X. The bone marrow-derived mesenchymal stem cells (BMSCs) alleviate diabetic peripheral neuropathy induced by STZ via activating GSK-3beta/beta-catenin signaling pathway. Environ Toxicol Pharmacol. 2020;79: 103432. - PubMed
-
- Boulton AJ, Kirsner RS, Vileikyte L. Clinical practice. Neuropathic diabetic foot ulcers. N Engl J Med. 2004;351(1):48–55. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

