The impact of ulinastatin on lymphocyte apoptosis and autophagy in sepsis patients
- PMID: 39567616
- PMCID: PMC11579385
- DOI: 10.1038/s41598-024-79878-y
The impact of ulinastatin on lymphocyte apoptosis and autophagy in sepsis patients
Abstract
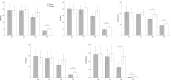
This study aimed to assess the influence of ulinastatin (UTI) on lymphocyte apoptosis and autophagy in sepsis patients, as well as its effect on inflammatory factors and vital organ function, with the goal of providing insights for improved clinical management of sepsis. A total of 40 sepsis patients were randomly assigned to the UTI group or the control group. The UTI group received standard treatment plus intravenous UTI, while the control group received standard treatment alone. Peripheral blood samples were collected at multiple time points for analysis of lymphocyte apoptosis, autophagy, inflammatory markers, and organ function. Various experimental techniques including Hoechst staining, transmission electron microscopy, and Western blot analysis were utilized to assess lymphocyte apoptosis, autophagy, and related protein expression levels. The study revealed that UTI treatment significantly inhibited lymphocyte apoptosis and promoted autophagy in sepsis patients. The levels of autophagy-related proteins LC3-II and Beclin-1 were substantially elevated, while the ratio of anti-apoptotic Bcl-2 to pro-apoptotic Bax was increased following UTI treatment. Furthermore, the levels of inflammatory markers IL-6, procalcitonin, and C-reactive protein were markedly reduced in the UTI group compared to the control group. Additionally, UTI treatment led to improved liver, kidney and cardiac function as evidenced by reduced levels of liver enzymes and creatinine, and cardiac enzymes. The findings of this study demonstrate that UTI exerts a protective effect on septic patients by inhibiting lymphocyte apoptosis, promoting autophagy, and attenuating systemic inflammation. Moreover, UTI treatment was associated with improved liver, kidney, and cardiac function in septic patients. These results contribute to a better understanding of the clinical management of sepsis and underscore the potential of UTI as a therapeutic intervention in septic patients.
Keywords: Autophagy; Inflammatory response; Lymphocyte apoptosis; Sepsis; Ulinastatin (UTI).
© 2024. The Author(s).
Conflict of interest statement
Declarations. Competing interests: The authors declare no competing interests. Ethics approval and consent to participate: The study protocol was approved by the Ethics Committee of the Second Affiliated Hospital of Harbin Medical University (KY2018-257). The study was performed in accordance with the ethical standards as laid out in the 1964 Declaration of Helsinki. Written informed consent was obtained from individual participants or their guardian.
Figures



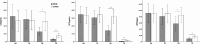
Similar articles
-
Ulinastatin improves survival of septic mice by suppressing inflammatory response and lymphocyte apoptosis.J Surg Res. 2013 Jun 15;182(2):296-302. doi: 10.1016/j.jss.2012.10.043. Epub 2012 Nov 9. J Surg Res. 2013. PMID: 23158408
-
Combination therapy of Ulinastatin with Thrombomodulin alleviates endotoxin (LPS) - induced liver and kidney injury via inhibiting apoptosis, oxidative stress and HMGB1/TLR4/NF-κB pathway.Bioengineered. 2022 Feb;13(2):2951-2970. doi: 10.1080/21655979.2021.2024686. Bioengineered. 2022. PMID: 35148668 Free PMC article.
-
Protective effect of rhubarb combined with ulinastatin for patients with sepsis.Medicine (Baltimore). 2020 Feb;99(7):e18895. doi: 10.1097/MD.0000000000018895. Medicine (Baltimore). 2020. PMID: 32049789 Free PMC article. Clinical Trial.
-
Autophagy Enhancing Contributes to the Organ Protective Effect of Alpha-Lipoic Acid in Septic Rats.Front Immunol. 2019 Jul 2;10:1491. doi: 10.3389/fimmu.2019.01491. eCollection 2019. Front Immunol. 2019. PMID: 31333648 Free PMC article.
-
Protective effect of Ulinastatin against murine models of sepsis: inhibition of TNF-α and IL-6 and augmentation of IL-10 and IL-13.Exp Toxicol Pathol. 2012 Sep;64(6):543-7. doi: 10.1016/j.etp.2010.11.011. Epub 2010 Dec 14. Exp Toxicol Pathol. 2012. PMID: 21159497
References
-
- van der Poll, T., Shankar-Hari, M. & Wiersinga, W. J. The immunology of sepsis. Immunity54, 2450–2464 (2021). - PubMed
-
- Matot, I. & Sprung, C. L. Definition of sepsis. Intensive Care Med.27, S3–S9 (2001). - PubMed
-
- Cassini, A. et al. Global report on the epidemiology and burden on sepsis: Current evidence, identifying gaps and future directions. In Global Report on the Epidemiology and Burden on Sepsis: Current Evidence, Identifying Gaps and Future Directions (2020).
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

