Mechanism of cold exposure delaying wound healing in mice
- PMID: 39568002
- PMCID: PMC11577949
- DOI: 10.1186/s12951-024-03009-y
Mechanism of cold exposure delaying wound healing in mice
Erratum in
-
Correction: Mechanism of cold exposure delaying wound healing in mice.J Nanobiotechnology. 2024 Dec 12;22(1):752. doi: 10.1186/s12951-024-03039-6. J Nanobiotechnology. 2024. PMID: 39663528 Free PMC article. No abstract available.
Abstract
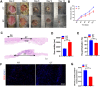
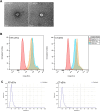
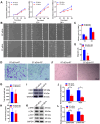
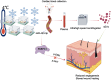
Cold temperatures have been shown to slow skin wound healing. However, the specific mechanisms underlying cold-induced impairment of wound healing remain unclear. Here, we demonstrate that small extracellular vesicles derived from cold-exposed mouse plasma (CT-sEVs) decelerate re-epithelialization, increase scar width, and weaken angiogenesis. CT-sEVs are enriched with miRNAs involved in the regulation of wound healing-related biological processes. Functional assays revealed that miR-423-3p, enriched in CT-sEVs, acts as a critical mediator in cold-induced impairment of angiogenic responses and poor wound healing by inhibiting phosphatase and poly(A) binding protein cytoplasmic 1 (PABPC1). These findings indicate that cold delays wound healing via miR-423-3p in plasma-derived sEVs through the inhibition of the ERK or AKT phosphorylation pathways. Our results enhance understanding of the molecular mechanisms by which cold exposure delays soft tissue wound healing.
Keywords: Angiogenesis; Cold exposure; Extracelluar Vesicles; PABPC1; Wound healing; miR-423-3p.
© 2024. The Author(s).
Conflict of interest statement
Declarations. Consent for publication: All authors agree for publication. Competing interests: The authors declare no competing interests.
Figures




Similar articles
-
Lactobacillus rhamnosus GG-derived extracellular vesicles promote wound healing via miR-21-5p-mediated re-epithelization and angiogenesis.J Nanobiotechnology. 2024 Oct 19;22(1):644. doi: 10.1186/s12951-024-02893-8. J Nanobiotechnology. 2024. PMID: 39427198 Free PMC article.
-
Exosomes from human umbilical cord blood accelerate cutaneous wound healing through miR-21-3p-mediated promotion of angiogenesis and fibroblast function.Theranostics. 2018 Jan 1;8(1):169-184. doi: 10.7150/thno.21234. eCollection 2018. Theranostics. 2018. PMID: 29290800 Free PMC article.
-
Extracellular vesicles-encapsulated microRNA-29b-3p from bone marrow-derived mesenchymal stem cells promotes fracture healing via modulation of the PTEN/PI3K/AKT axis.Exp Cell Res. 2022 Mar 15;412(2):113026. doi: 10.1016/j.yexcr.2022.113026. Epub 2022 Jan 10. Exp Cell Res. 2022. PMID: 35026284
-
Extracellular vesicle-enclosed miR-486-5p mediates wound healing with adipose-derived stem cells by promoting angiogenesis.J Cell Mol Med. 2020 Sep;24(17):9590-9604. doi: 10.1111/jcmm.15387. Epub 2020 Jul 14. J Cell Mol Med. 2020. PMID: 32666704 Free PMC article.
-
Endothelial cell-derived small extracellular vesicles suppress cutaneous wound healing through regulating fibroblasts autophagy.Clin Sci (Lond). 2019 May 2;133(9):CS20190008. doi: 10.1042/CS20190008. Print 2019 May 15. Clin Sci (Lond). 2019. PMID: 30988132
References
-
- Bukowiecki LJ. Energy balance and diabetes. The effects of cold exposure, exercise training, and diet composition on glucose tolerance and glucose metabolism in rat peripheral tissues. Can J Physiol Pharmacol. 1989;67(4):382–93. - PubMed
-
- Gostimirovic M, Novakovic R, Rajkovic J, et al. The influence of climate change on human cardiovascular function. Arch Environ Occup Health. 2020;75(7):406–14. - PubMed
-
- Urbański A, Czarniewska E, Baraniak E, et al. Impact of cold on the immune system of burying beetle, Nicrophorus vespilloides (Coleoptera: Silphidae). Insect Sci. 2017;24(3):443–54. - PubMed
MeSH terms
Substances
Grants and funding
- 82370892/the National Natural Science Foundation of China
- 2021YFC2501701/National Key Research & Development Program
- Z2023026/National Clinical Key Specialties Major Research Projects
- 20231696/The Health Research Project in Hunan Province
- W20243019/Health Research Project of Hunan Provincial Health Commission
LinkOut - more resources
Full Text Sources
Miscellaneous

