Long term outcomes of intracarotid arterial transfusion of circulatory-derived autologous CD34 + cells for acute ischemic stroke patients-A randomized, open-label, controlled phase II clinical trial
- PMID: 39568005
- PMCID: PMC11577584
- DOI: 10.1186/s13287-024-04021-7
Long term outcomes of intracarotid arterial transfusion of circulatory-derived autologous CD34 + cells for acute ischemic stroke patients-A randomized, open-label, controlled phase II clinical trial
Abstract
Background: This phase II randomized controlled trial tested whether the intracarotid arterial administration (ICAA) of autologous CD34 + cells to patients within 14 ± 7 days after acute ischemic stroke (IS) could be safe and further improve short- and long-term outcomes.
Methods: Between January 2018 and March 2022, 28 consecutive patients were equally randomly allocated to the cell-treated group (CD34 + cells/3.0 × 107/patient) or the control group (receiving optimal medical therapy). CD34 + cells were transfused into the ipsilateral brain infarct zone of cell-treated patients via the ICAA in the catheterization room.
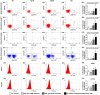
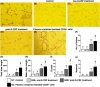
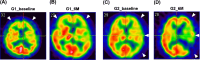
Results: The results demonstrated 100% safety and success rates for the procedure, and no long-term tumorigenesis was observed in cell-treated patients. In cell-treated patients, the angiogenesis capacity of circulating endothelial progenitor cells (EPCs)/Matrigel was significantly greater after treatment than before treatment with granulocyte colony-stimulating factor (all p < 0.001). Blood samples from the right internal jugular vein of the cell-treated patients presented significantly greater levels of the stromal cell-derived factor 1α/EPC at 5, 10 and 30 min compared with 0 min (all p < 0.005). The National Institute of Health Stroke Scale scores were similar upon presentation, but a greater response was observed by Days 30 and 90 in the cell-treated group than in the control group. Tc-99 m brain perfusion was significantly greater at 180 days in the cell-treated group than in the control group (p = 0.046). The combined long-term end points (defined as death/recurrent stroke/or severe disability) were notably lower in the control group compared with the cell-treated group (14.3% vs. 50.0%, p = 0.103).
Conclusion: Intracarotid transfusion of autologous CD34 + cells is safe and might improve long-term outcomes in patients with acute IS. Trial registration ISRCTN, ISRCTN15677760. Registered 23 April 2018- Retrospectively registered, https://doi.org/10.1186/ISRCTN15677760.
Keywords: Acute ischemic stroke; Angiogenesis; Endothelial progenitor cells; Intracarotid transfusion of CD34 + cells; Neurological outcomes.
© 2024. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: This clinical trial protocol was reviewed and approved by the Taiwan Food and Drug Administration (Approval number: 1109012692) and the Institutional Review Board of Chang Gung Medical Foundation (Approval number: 201700116A0C502). This project entitled “An investigation of the therapeutic impact of intra-carotid arterial transfusion of autologous peripheral blood-derived stem cell/progenitor cell (CD34 +) therapy on brain ischemic stroke—a phase II clinical trial” was approved on July 17, 2017. Furthermore, all patients signed the approved informed consent form, and all the procedures were performed in accordance with the principles of the Declaration of Helsinki. Trial registration: ISRCTN, ISRCTN15677760. Registered 23 April 2018- Retrospectively registered, https://doi.org/ https://doi.org/10.1186/ISRCTN15677760 . Consent for publication: Not applicable. Competing interests: The authors declare that they have no competing interests.
Figures


References
-
- Krishnamurthi RV, Moran AE, Feigin VL, Barker-Collo S, Norrving B, Mensah GA, Taylor S, Naghavi M, Forouzanfar MH, Nguyen G, Johnson CO. Stroke prevalence, mortality and disability-adjusted life years in adults aged 20–64 years in 1990–2013: data from the global burden of disease 2013 study. Neuroepidemiology. 2015;45(3):190–202. - PubMed
-
- Krishnamurthi RV, Ikeda T, Feigin VL. Global, regional and country-specific burden of ischaemic stroke, intracerebral haemorrhage and subarachnoid haemorrhage: a systematic analysis of the global burden of disease study 2017. Neuroepidemiology. 2020;54:171–9. - PubMed
-
- Feigin VL, Brainin M, Norrving B, Martins S, Sacco RL, Hacke W, Fisher M, Pandian J, Lindsay P. World stroke organization (WSO): global stroke fact sheet 2022. Int J Stroke. 2022;17:18–29. - PubMed
-
- Pessin MS, Duncan GW, Mohr JP, Poskanzer DC. Clinical and angiographic features of carotid transient ischemic attacks. N Engl J Med. 1977;296:358–62. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

