Dual ligand-enabled iron and halogen-containing carboxylate-based photocatalysis for chloro/fluoro-polyhaloalkylation of alkenes
- PMID: 39568912
- PMCID: PMC11575577
- DOI: 10.1039/d4sc04038d
Dual ligand-enabled iron and halogen-containing carboxylate-based photocatalysis for chloro/fluoro-polyhaloalkylation of alkenes
Abstract
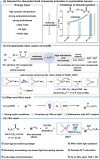
Herein, we demonstrate a practical dual ligand-enabled iron photocatalysis paradigm-converting all kinds of halogen-containing carboxylates (C n X m COO-, X: F, Cl, Br) into C n X m radicals for the valuable chloro/fluoro-polyhaloalkylation of non-activated alkenes with easily available trichloroacetonitrile/Selectfluor as the electrophilic halogenation reagent. The modular in situ assembly of the effective iron and C n X m COO--based light-harvesting species using the two ligands-OMe/CF3-substituted bipyridine and acetonitrile/trichloroacetonitrile is evidenced by detailed mechanistic studies. The late-stage modification, low loading amount of iron (TON: 257) and feasible gram-scale synthesis show the utility of this protocol. We thus anticipate that the dual ligand-enabled iron photocatalysis paradigm may facilitate activation and transformation of inert bulk chemicals.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures



Similar articles
-
Iron photocatalysis via Brønsted acid-unlocked ligand-to-metal charge transfer.Nat Commun. 2024 Jul 20;15(1):6115. doi: 10.1038/s41467-024-50507-6. Nat Commun. 2024. PMID: 39033136 Free PMC article.
-
Three-Component Perfluoroalkylvinylation of Alkenes Enabled by Dual DBU/Fe Catalysis.Chemistry. 2023 Feb 7;29(8):e202203332. doi: 10.1002/chem.202203332. Epub 2022 Dec 14. Chemistry. 2023. PMID: 36351885
-
Iron-Catalyzed Halogen Exchange of Trifluoromethyl Arenes*.Chemistry. 2021 Jul 26;27(42):10839-10843. doi: 10.1002/chem.202101324. Epub 2021 Jun 17. Chemistry. 2021. PMID: 34137084
-
Halogen bond-induced electrophilic aromatic halogenations.Org Biomol Chem. 2021 Sep 15;19(35):7518-7534. doi: 10.1039/d1ob00936b. Org Biomol Chem. 2021. PMID: 34346463 Review.
-
Electrophilic Iron Catalyst Paired with a Lithium Cation Enables Selective Functionalization of Non-Activated Aliphatic C-H Bonds via Metallocarbene Intermediates.Angew Chem Int Ed Engl. 2019 Sep 23;58(39):13904-13911. doi: 10.1002/anie.201905986. Epub 2019 Aug 21. Angew Chem Int Ed Engl. 2019. PMID: 31338944 Review.
References
-
- Prier C. K. Rankic D. A. MacMillan D. W. Chem. Rev. 2013;113:5322–5363. doi: 10.1021/cr300503r. - DOI - PMC - PubMed
- Bellotti P. Huang H.-M. Faber T. Glorius F. Chem. Rev. 2023;123:4237–4352. doi: 10.1021/acs.chemrev.2c00478. - DOI - PubMed
- Hartwig J. F. Larsen M. A. ACS Cent. Sci. 2016;2:281–292. doi: 10.1021/acscentsci.6b00032. - DOI - PMC - PubMed
- Jiang H. Studer A. Chem. Soc. Rev. 2020;49:1790–1811. doi: 10.1039/C9CS00692C. - DOI - PubMed
- Narayanam J. M. Stephenson C. R. Chem. Soc. Rev. 2011;40:102–113. doi: 10.1039/B913880N. - DOI - PubMed
- Ravelli D. Protti S. Fagnoni M. Chem. Rev. 2016;116:9850–9913. doi: 10.1021/acs.chemrev.5b00662. - DOI - PubMed
- Holmberg-Douglas N. Nicewicz D. A. Chem. Rev. 2022;122:1925–2016. doi: 10.1021/acs.chemrev.1c00311. - DOI - PMC - PubMed
-
- Cho E. J. Senecal T. D. Kinzel T. Zhang Y. Watson D. A. Buchwald S. L. Science. 2010;328:1679–1681. doi: 10.1126/science.1190524. - DOI - PMC - PubMed
- Rodríguez N. Goossen L. J. Chem. Soc. Rev. 2011;40:5030–5048. doi: 10.1039/C1CS15093F. - DOI - PubMed
- Weaver J. D. Recio A. Grenning A. J. Tunge J. A. Chem. Rev. 2011;111:1846–1913. doi: 10.1021/cr1002744. - DOI - PMC - PubMed
- Wei Y. Hu P. Zhang M. Su W. Chem. Rev. 2017;117:8864–8907. doi: 10.1021/acs.chemrev.6b00516. - DOI - PubMed
- Li X. Yang T. Li J. Li X. Chen P. Lin Z. Liu G. Nat. Chem. 2023;15:862–871. doi: 10.1038/s41557-023-01192-3. - DOI - PubMed
-
- Fujiwara Y. Dixon J. A. O'Hara F. Funder E. D. Dixon D. D. Rodriguez R. A. Baxter R. D. Herlé B. Sach N. Collins M. R. Ishihara Y. Baran P. S. Nature. 2012;492:95–99. doi: 10.1038/nature11680. - DOI - PMC - PubMed
- Yin D. Su D. Jin J. Cell Rep. Phys. Sci. 2020;1:100141. doi: 10.1016/j.xcrp.2020.100141. - DOI
- Tang M. Draper F. Pham L. N. Ho C. C. Huang H. Sun J. Thickett S. C. Coote M. L. Connell T. U. Bissember A. C. J. Org. Chem. 2024;89:2683–2690. doi: 10.1021/acs.joc.3c02746. - DOI - PubMed
-
- Lai C. Mallouk T. E. J. Chem. Soc., Chem. Commun. 1993:1359–1361. doi: 10.1039/C39930001359. - DOI
- Liu W. Yang X. Zhou Z.-Z. Li C.-J. Chem. 2017;2:688–702. doi: 10.1016/j.chempr.2017.03.009. - DOI
- Yuan T.-T. Chen J. Pham L. N. Paul S. White L. V. Li J. Lan P. Coote M. L. Banwell M. G. He Y.-T. Org. Chem. Front. 2023;10:4649–4657. doi: 10.1039/D3QO00754E. - DOI
LinkOut - more resources
Full Text Sources

