Influenza, COVID-19, and Respiratory Syncytial Virus Vaccination Coverage Among Adults - United States, Fall 2024
- PMID: 39570786
- PMCID: PMC11581203
- DOI: 10.15585/mmwr.mm7346a1
Influenza, COVID-19, and Respiratory Syncytial Virus Vaccination Coverage Among Adults - United States, Fall 2024
Abstract
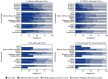
The Advisory Committee on Immunization Practices (ACIP) recommends annual influenza and COVID-19 vaccination for all persons aged ≥6 months, including adults aged ≥18 years. ACIP also recommends a single lifetime dose of respiratory syncytial virus (RSV) vaccine for adults aged ≥75 years and for those aged 60-74 years who are at increased risk for severe RSV disease. Data from the National Immunization Survey-Adult COVID Module, a random-digit-dialed cellular telephone survey of U.S. adults aged ≥18 years, are used to monitor influenza, COVID-19, and RSV vaccination coverage. By the week ending November 9, 2024, an estimated 34.7% of adults aged ≥18 years reported having received an influenza vaccine, and 17.9% reported having received a COVID-19 vaccine for the 2024-25 respiratory virus season; 39.7% of adults aged ≥75 years, and 31.6% of adults aged 60-74 years at increased risk for severe RSV, had ever received an RSV vaccine. Coverage varied by jurisdiction and demographic characteristics and was lowest among younger adults and those without health insurance. Although early season estimates indicate that many adults are unprotected from respiratory virus infections, many appeared open to vaccination: overall, approximately 35% and 41% of adults aged ≥18 years reported that they definitely or probably will receive or were unsure about receiving influenza and COVID-19 vaccines, respectively, and 40% of adults aged ≥75 years reported that they definitely or probably will receive or were unsure about receiving RSV vaccine. Health care providers and immunization programs still have time to expand outreach activities and promote vaccination to increase coverage in preparation for the height of the respiratory virus season. Using these data can help health care providers and immunization programs identify undervaccinated populations and understand vaccination patterns to guide planning, implementation, and evaluation of vaccination activities.
Conflict of interest statement
All authors have completed and submitted the International Committee of Medical Journal Editors form for disclosure of potential conflicts of interest. No potential conflicts of interest were disclosed.
Figures

References
-
- Grohskopf LA, Ferdinands JM, Blanton LH, Broder KR, Loehr J. Prevention and control of seasonal influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices—United States, 2024–25 influenza season. MMWR Recomm Rep 2024;73:1–25. 10.15585/mmwr.rr7305a1 - DOI - PMC - PubMed
-
- Black CL, O’Halloran A, Hung MC, et al.; Influenza-Associated Hospitalization Surveillance Network. Vital signs: influenza hospitalizations and vaccination coverage by race and ethnicity—United States, 2009–10 through 2021–22 influenza seasons. MMWR Morb Mortal Wkly Rep 2022;71:1366–73. 10.15585/mmwr.mm7143e1 - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

